Presented here, is an extensive 35 parameter experimental data set of a cylindrical 21700 commercial cell (LGM50), for an electrochemical pseudo-two-dimensional (P2D) model. The experimental methodologies for tear-down and subsequent chemical, physical, electrochemical kinetics and thermodynamic analysis, and their accuracy and validity are discussed. Chemical analysis of the LGM50 cell shows that it is comprised of a NMC 811 positive electrode and bi-component Graphite-SiOx negative electrode. The thermodynamic open circuit voltages (OCV) and lithium stoichiometry in the electrode are obtained using galvanostatic intermittent titration technique (GITT) in half cell and three-electrode full cell configurations. The activation energy and exchange current coefficient through electrochemical impedance spectroscopy (EIS) measurements. Apparent diffusion coefficients are estimated using the Sand equation on the voltage transient during the current pulse; an expansion factor was applied to the bi-component negative electrode data to reflect the average change in effective surface area during lithiation. The 35 parameters are applied within a P2D model to show the fit to experimental validation LGM50 cell discharge and relaxation voltage profiles at room temperature. The accuracy and validity of the processes and the techniques in the determination of these parameters are discussed, including opportunities for further modelling and data analysis improvements.

The Electrochemical Society (ECS) was founded in 1902 to advance the theory and practice at the forefront of electrochemical and solid state science and technology, and allied subjects.
ISSN: 1945-7111
JES is the flagship journal of The Electrochemical Society. Published continuously from 1902 to the present, JES remains one of the most highly-cited journals in electrochemistry and solid-state science and technology.
Chang-Hui Chen et al 2020 J. Electrochem. Soc. 167 080534
Eniko S. Zsoldos et al 2024 J. Electrochem. Soc. 171 080527
Lithium iron phosphate (LFP) battery cells are ubiquitous in electric vehicles and stationary energy storage because they are cheap and have a long lifetime. This work compares LFP/graphite pouch cells undergoing charge-discharge cycles over five state of charge (SOC) windows (0%–25%, 0%–60%, 0%–80%, 0%–100%, and 75%–100%). Cycling LFP cells across a lower average SOC results in less capacity fade than cycling across a higher average SOC, regardless of depth of discharge. The primary capacity fade mechanism is lithium inventory loss due to: lithiated graphite reactivity with electrolyte, which increases incrementally with SOC, and lithium alkoxide species causing iron dissolution and deposition on the negative electrode at high SOC which further accelerates lithium inventory loss. Our results show that even low voltage LFP systems (3.65 V) have a tradeoff between average SOC and lifetime. Operating LFP cells at lower average SOC can extend their lifetime substantially in both EV and grid storage applications.
Peter Keil et al 2016 J. Electrochem. Soc. 163 A1872
In this study, the calendar aging of lithium-ion batteries is investigated at different temperatures for 16 states of charge (SoCs) from 0 to 100%. Three types of 18650 lithium-ion cells, containing different cathode materials, have been examined. Our study demonstrates that calendar aging does not increase steadily with the SoC. Instead, plateau regions, covering SoC intervals of more than 20%–30% of the cell capacity, are observed wherein the capacity fade is similar. Differential voltage analyses confirm that the capacity fade is mainly caused by a shift in the electrode balancing. Furthermore, our study reveals the high impact of the graphite electrode on calendar aging. Lower anode potentials, which aggravate electrolyte reduction and thus promote solid electrolyte interphase growth, have been identified as the main driver of capacity fade during storage. In the high SoC regime where the graphite anode is lithiated more than 50%, the low anode potential accelerates the loss of cyclable lithium, which in turn distorts the electrode balancing. Aging mechanisms induced by high cell potential, such as electrolyte oxidation or transition-metal dissolution, seem to play only a minor role. To maximize battery life, high storage SoCs corresponding to low anode potential should be avoided.
Yuliya Preger et al 2020 J. Electrochem. Soc. 167 120532
Energy storage systems with Li-ion batteries are increasingly deployed to maintain a robust and resilient grid and facilitate the integration of renewable energy resources. However, appropriate selection of cells for different applications is difficult due to limited public data comparing the most commonly used off-the-shelf Li-ion chemistries under the same operating conditions. This article details a multi-year cycling study of commercial LiFePO4 (LFP), LiNixCoyAl1−x−yO2 (NCA), and LiNixMnyCo1−x−yO2 (NMC) cells, varying the discharge rate, depth of discharge (DOD), and environment temperature. The capacity and discharge energy retention, as well as the round-trip efficiency, were compared. Even when operated within manufacturer specifications, the range of cycling conditions had a profound effect on cell degradation, with time to reach 80% capacity varying by thousands of hours and cycle counts among cells of each chemistry. The degradation of cells in this study was compared to that of similar cells in previous studies to identify universal trends and to provide a standard deviation for performance. All cycling files have been made publicly available at batteryarchive.org, a recently developed repository for visualization and comparison of battery data, to facilitate future experimental and modeling efforts.
Jorn M. Reniers et al 2019 J. Electrochem. Soc. 166 A3189
The maximum energy that lithium-ion batteries can store decreases as they are used because of various irreversible degradation mechanisms. Many models of degradation have been proposed in the literature, sometimes with a small experimental data set for validation. However, a comprehensive comparison between different model predictions is lacking, making it difficult to select modelling approaches which can explain the degradation trends actually observed from data. Here, various degradation models from literature are implemented within a single particle model framework and their behavior is compared. It is shown that many different models can be fitted to a small experimental data set. The interactions between different models are simulated, showing how some of the models accelerate degradation in other models, altering the overall degradation trend. The effects of operating conditions on the various degradation models is simulated. This identifies which models are enhanced by which operating conditions and might therefore explain specific degradation trends observed in data. Finally, it is shown how a combination of different models is needed to capture different degradation trends observed in a large experimental data set. Vice versa, only a large data set enables to properly select the models which best explain the observed degradation.
A. K. Padhi et al 1997 J. Electrochem. Soc. 144 1188
Reversible extraction of lithium from 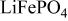 (triphylite) and insertion of lithium into
(triphylite) and insertion of lithium into 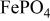 at 3.5 V vs. lithium at 0.05 mA/cm2 shows this material to be an excellent candidate for the cathode of a low‐power, rechargeable lithium battery that is inexpensive, nontoxic, and environmentally benign. Electrochemical extraction was limited to ∼0.6 Li/formula unit; but even with this restriction the specific capacity is 100 to 110 mAh/g. Complete extraction of lithium was performed chemically; it gave a new phase,
at 3.5 V vs. lithium at 0.05 mA/cm2 shows this material to be an excellent candidate for the cathode of a low‐power, rechargeable lithium battery that is inexpensive, nontoxic, and environmentally benign. Electrochemical extraction was limited to ∼0.6 Li/formula unit; but even with this restriction the specific capacity is 100 to 110 mAh/g. Complete extraction of lithium was performed chemically; it gave a new phase, 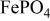 , isostructural with heterosite,
, isostructural with heterosite, 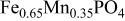 . The
. The 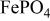 framework of the ordered olivine
framework of the ordered olivine 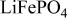 is retained with minor displacive adjustments. Nevertheless the insertion/extraction reaction proceeds via a two‐phase process, and a reversible loss in capacity with increasing current density appears to be associated with a diffusion‐limited transfer of lithium across the two‐phase interface. Electrochemical extraction of lithium from isostructural
is retained with minor displacive adjustments. Nevertheless the insertion/extraction reaction proceeds via a two‐phase process, and a reversible loss in capacity with increasing current density appears to be associated with a diffusion‐limited transfer of lithium across the two‐phase interface. Electrochemical extraction of lithium from isostructural 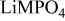 (M = Mn, Co, or Ni) with an
(M = Mn, Co, or Ni) with an 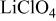 electrolyte was not possible; but successful extraction of lithium from
electrolyte was not possible; but successful extraction of lithium from 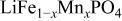 was accomplished with maximum oxidation of the
was accomplished with maximum oxidation of the 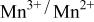 occurring at x = 0.5. The
occurring at x = 0.5. The 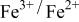 couple was oxidized first at 3.5 V followed by oxidation of the
couple was oxidized first at 3.5 V followed by oxidation of the 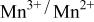 couple at 4.1 V vs. lithium. The
couple at 4.1 V vs. lithium. The 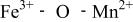 interactions appear to destabilize the
interactions appear to destabilize the 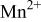 level and stabilize the
level and stabilize the 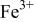 level so as to make the
level so as to make the 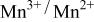 energy accessible.
energy accessible.
E. Peled and S. Menkin 2017 J. Electrochem. Soc. 164 A1703
The Solid-Electrolyte-Interphase (SEI) model for non-aqueous alkali-metal batteries constitutes a paradigm change in the understanding of lithium batteries and has thus enabled the development of safer, durable, higher-power and lower-cost lithium batteries for portable and EV applications. Prior to the publication of the SEI model (1979), researchers used the Butler-Volmer equation, in which a direct electron transfer from the electrode to lithium cations in the solution is assumed. The SEI model proved that this is a mistaken concept and that, in practice, the transfer of electrons from the electrode to the solution in a lithium battery, must be prevented, since it will result in fast self-discharge of the active materials and poor battery performance. This model provides [E. Peled, in “Lithium Batteries,” J.P. Gabano (ed), Academic Press, (1983), E. Peled, J. Electrochem. Soc., 126, 2047 (1979).] new equations for: electrode kinetics (io and b), anode corrosion, SEI resistivity and growth rate and irreversible capacity loss of lithium-ion batteries. This model became a cornerstone in the science and technology of lithium batteries. This paper reviews the past, present and the future of SEI batteries.
Weilong Ai et al 2020 J. Electrochem. Soc. 167 013512
Whilst extensive research has been conducted on the effects of temperature in lithium-ion batteries, mechanical effects have not received as much attention despite their importance. In this work, the stress response in electrode particles is investigated through a pseudo-2D model with mechanically coupled diffusion physics. This model can predict the voltage, temperature and thickness change for a lithium cobalt oxide-graphite pouch cell agreeing well with experimental results. Simulations show that the stress level is overestimated by up to 50% using the standard pseudo-2D model (without stress enhanced diffusion), and stresses can accelerate the diffusion in solid phases and increase the discharge cell capacity by 5.4%. The evolution of stresses inside electrode particles and the stress inhomogeneity through the battery electrode have been illustrated. The stress level is determined by the gradients of lithium concentration, and large stresses are generated at the electrode-separator interface when high C-rates are applied, e.g. fast charging. The results can explain the experimental results of particle fragmentation close to the separator and provide novel insights to understand the local aging behaviors of battery cells and to inform improved battery control algorithms for longer lifetimes.
D. Bernardi et al 1985 J. Electrochem. Soc. 132 5
A general energy balance for battery systems has been developed. This equation is useful for estimating cell thermal characteristics. Reliable predictions of cell temperature and heat‐generation rate are required for the design and thermal management of battery systems. The temperature of a cell changes as a result of electrochemical reactions, phase changes, mixing effects, and joule heating. The equation developed incorporates these effects in a complete and general manner. Simplifications and special cases are discussed. The results of applying the energy balance to a mathematical model of the  cell discharged through two different reaction mechanisms are given as examples. The examples illustrate how the energy equation may be applied to a specific system to examine the relative contributions corresponding to the terms in the equation. The examples show that the processes involved in cell heat generation may be complex and that the application of a sufficiently general energy equation is advantageous.
cell discharged through two different reaction mechanisms are given as examples. The examples illustrate how the energy equation may be applied to a specific system to examine the relative contributions corresponding to the terms in the equation. The examples show that the processes involved in cell heat generation may be complex and that the application of a sufficiently general energy equation is advantageous.
Manuel Ank et al 2023 J. Electrochem. Soc. 170 120536
Battery research depends upon up-to-date information on the cell characteristics found in current electric vehicles, which is exacerbated by the deployment of novel formats and architectures. This necessitates open access to cell characterization data. Therefore, this study examines the architecture and performance of first-generation Tesla 4680 cells in detail, both by electrical characterization and thermal investigations at cell-level and by disassembling one cell down to the material level including a three-electrode analysis. The cell teardown reveals the complex cell architecture with electrode disks of hexagonal symmetry as well as an electrode winding consisting of a double-sided and homogeneously coated cathode and anode, two separators and no mandrel. A solvent-free anode fabrication and coating process can be derived. Energy-dispersive X-ray spectroscopy as well as differential voltage, incremental capacity and three-electrode analysis confirm a NMC811 cathode and a pure graphite anode without silicon. On cell-level, energy densities of 622.4 Wh/L and 232.5 Wh/kg were determined while characteristic state-of-charge dependencies regarding resistance and impedance behavior are revealed using hybrid pulse power characterization and electrochemical impedance spectroscopy. A comparatively high surface temperature of ∼70 °C is observed when charging at 2C without active cooling. All measurement data of this characterization study are provided as open source.
Yohei Nagatomo et al 2026 J. Electrochem. Soc. 173 114508
A unified theoretical framework is proposed for describing the exchange current density of electrode reactions in solid oxide cells, covering charge transfer, dissociative adsorption, and surface transport as possible rate-determining steps. By combining kinetic processes with equilibrium relations among dissociative adsorption equilibria, the apparent dependencies of exchange current density on gas partial pressures are systematically derived, together with general forms that also accommodate temperature dependence. The apparent activation energy can be expressed as the sum of the intrinsic barrier of the rate-determining step and the standard enthalpies of quasi-equilibrated reactions, weighted by surface coverage. Apparent reaction orders are likewise formulated as the combination of kinetic and thermodynamic contributions, revealing fundamental constraints on their accessible values. As a concrete example, analysis of the H2/H2O system demonstrates that the possible combinations of reaction orders fall within a parallelogram-shaped region defined by ideal Langmuir adsorption equilibria. This result illustrates how this framework not only explains empirical power-law relations but also helps clarify why distinct elementary mechanisms can yield indistinguishable pressure dependences. This approach provides a consistent basis for interpreting electrode kinetics and offers a general tool for linking microscopic reaction models with macroscopic experimental observables in solid oxide cells.
figure placeholder
Highlights
Explains gas-pressure dependencies of exchange current in solid oxide cells.
Dependencies combine kinetic effects with thermodynamics via surface coverage.
H2/H2O reaction places apparent pressure exponents inside a parallelogram region.
Highlights non-uniqueness of pressure exponents across elementary mechanisms.
Rajan Maurya et al 2026 J. Electrochem. Soc. 173 114507
The effect of pre-wetting on the catalyst utilization and durability of high-temperature proton exchange membrane fuel cell (HT-PEMFC) gas diffusion electrodes (GDEs) was investigated in a three-electrode configuration. Pre-wetting protocols included treatment in ethanol-water mixture (0, 10, 50, and 100% v/v ethanol), ethanol-water vapor, and concentrated orthophosphoric acid (14.85 M) at 150 °C. All pre-wetting approaches significantly enhanced electrode wettability by reducing hydrophobicity and surface tension, thereby improving initial catalyst utilization. However, accelerated stress test (AST) revealed a trade-off between utilization and durability, with wetted electrodes exhibiting lower electrochemically active surface area and Pt retention than untreated electrodes. Changes in Pt crystallite and particle sizes during the AST were estimated through X-ray diffraction analysis and transmission electron microscopy. All three wetting procedures have similar effects on catalyst utilization and durability without compromising the structural integrity. Single-cell measurements showed that while pre-wetting is effective for rapid screening and improving the initial performance, its benefits are not directly transferable to practical operation without further optimization. These findings highlight the trade-off between catalyst utilization and durability in HT-PEMFC GDEs.
figure placeholder
Highlights
Proper wetting of HT-PEMFC electrode in aqueous electrolytes is challenging
Methods to improve wettability (alcoholic solvent/temperature) are suggested as protocols
Initial activity and durability of the electrodes depend strongly on the wetting method
Qunying Kang et al 2026 J. Electrochem. Soc. 173 110516
Li-O2 batteries have attracted widespread attention due to their high theoretical energy density, but their practical application is limited by the slow kinetics and high overpotentials of the ORR and OER. Developing efficient electrocatalysts is key to overcoming this bottleneck. In this study, we successfully synthesized a Co, N, and C-doped RuO2/MnO2 composite catalyst via ultrasound-assisted hydrothermal synthesis and vacuum filtration, and subsequently formed a Co-N-C-RuO2/MnO2 composite cathode on a carbon paper substrate. Through systematic material characterization, electrochemical testing, and microscopic analysis of the reaction mechanism, we investigated the catalytic performance of this composite cathode in Li-O2 batteries. The results indicate that, compared to the single RuO2/MnO2 cathode, the Co-N-C-RuO2/MnO2 composite cathode exhibits lower charge-discharge overpotentials, a 33% increase in discharge specific capacity, and a 77% improvement in cycling stability. Mechanistic analysis shows that the doped Co, N, and C elements significantly enhance the cathode’s catalytic activity by increasing active sites and optimizing the electronic structure, thereby promoting the nucleation and reversible decomposition of Li2O2, and improving overall battery performance.
figure placeholder
Highlights
A novel formulation of the Co-N-C-RuO2/MnO2 catalyst has been proposed.
There is a good synergistic effect between Co-N-C and RuO2/MnO2.
The catalytic activity and reaction mechanism are key factors affecting battery performance.
Chenxi Liu et al 2026 J. Electrochem. Soc. 173 111503
Critical chloride concentration (Cth) for depassivation is an essential parameter for ensuring the durability of reinforced concrete structures, while the mechanism by which chlorides initiate the failure of the passive film at this threshold remains debated. Cth required by passive film breakdown was determined by a cyclic potentiodynamic polarization test here. A combination of Mott-Schottky test and X-ray photoelectron spectroscopy was further applied to explore the semiconductive properties and chemical composition of the film under this critical environment. The results show that this Cth is lower than that determined by a sharp change in corrosion potential, polarization resistance, or corrosion current density. Neither the pitting potential nor the cation vacancy concentration is sensitive to chloride near this threshold. The role of chloride is to accelerate the passive film dissolution, causing the film to thin and eventually break down. It does not penetrate the film or even reach the metal substrate.
figure placeholder
Highlights
Cl− accelerates the dissolution of the passive film but does not penetrate it.
Cf/s, and Epit are not sensitive to Cl−in highly alkaline SCPSs.
Cth was determined by the cyclic potentiodynamic polarization test.
This Cth is lower than the one determined by a sharp change of Ecorr, Rp, or icorr.
Chunmei Wang et al 2026 J. Electrochem. Soc. 173 110515
Sodium vanadate (NaVO3) has attracted increasing attention as an anode material for sodium-ion batteries owing to its high theoretical capacity. However, its practical application is severely hindered by poor electronic conductivity and pronounced structural instability during cycling. Here we report an interconnected NaVO3–carbon nanotube (CNT) hybrid architecture in which NaVO3 nanostructures are in situ grown along a multi-walled CNT framework via an evaporation-induced solution process. This rationally designed composite establishes continuous electron-transport pathways while mechanically reinforcing the active material against sodiation-induced stress. Comprehensive structural and spectroscopic analyses reveal strong interfacial electronic coupling between NaVO3 and CNTs, accompanied by the formation of a hierarchical porous network that facilitates electrolyte infiltration and rapid Na+ diffusion. Although the CNT framework improves the electronic conductivity, Na+ transport kinetics, and initial reversibility of NaVO3, the remaining capacity decay indicates that irreversible structural reconstruction still represents a major limitation for NaVO3-based anodes. In situ X-ray diffraction further uncovers an irreversible crystalline-to-amorphous transition during the first sodiation process, highlighting the critical role of CNT confinement in stabilizing the amorphous NaVO3 framework upon cycling. Therefore, this work provides mechanistic insight into the conductivity enhancement and structural degradation behavior of NaVO3, offering useful guidance for the future design of structurally stabilized sodium vanadate anodes.
figure placeholder
Ridge M. Bachman et al 2026 J. Electrochem. Soc. 173 110514
The transition to new energy resources is driving the need for efficient energy storage. Redox flow batteries (RFBs) are a promising solution due to their flexible design and safer materials, particularly for long-duration storage applications. Despite these advantages, RFB technologies still have considerable room for growth in terms of utilizing all of their available energy storage density. Herein, we assess RFB energy storage density utilization trends for multiple chemistries by accounting for their unique thermodynamic limits, energy efficiencies, and charge capacity constraints. Of the RFBs chemistries analyzed, most still use less than 60% of their theoretical energy storage capacity. The conventional all-vanadium RFB was the closest to its thermodynamic limit available. Moreover, the volumetric footprint and electrolyte cost analyses demonstrate the real-world implications of these inefficiencies. This analysis further indicates that future studies would benefit from clearer reporting of two key quantities: discharge efficiency, which is more directly tied to practically recoverable energy density than round-trip energy efficiency, and state-of-charge or capacity-utilization limits, which govern access to theoretical storage capacity. More consistent reporting of these factors would improve the accuracy and comparability of energy storage utilization analyses across RFB chemistries.
figure placeholder
Highlights
Energy storage density values from energy efficiency and capacity constraints
Redox flow batteries use less than 60% of their energy storage density limit
Inefficiencies in redox flow batteries increase their volumetric footprint
Cost effective electrolytes make up for underutilized energy storage density
Talal F. Qahtan et al 2026 J. Electrochem. Soc. 173 110506
Starting batteries are critical electrochemical energy storage systems that deliver high power pulses for engine ignition and auxiliary electrical functions. Their performance, durability, and safety are governed by complex electrochemical interactions involving electrode materials, electrolytes, interfacial processes, and operating conditions. This review provides a comprehensive electrochemical perspective on major starting battery technologies, including flooded lead–acid, absorbed glass mat, gel cell, lithium-ion, and nickel–cadmium batteries. Key electrochemical performance-limiting factors such as charge–discharge behavior, polarization losses, internal resistance, ion transport, and electrode–electrolyte interfacial stability are systematically analyzed. Further, the review emphasizes degradation and aging mechanisms such as sulfation, grid corrosion, gas evolution, lithium plating, and thermal runaway. It explains their electrochemical origins and how they affect long-term battery stability. The effects of temperature, charge rate, discharge rate, and material composition on electrochemical efficiency and cycle life are critically discussed. Recent advances in electrode materials, electrolyte systems (gel, polymer, ionic liquid, and deep eutectic electrolytes), and separator design are reviewed from an electrochemical perspective. These advances focus on strategies to enhance charge-transfer kinetics, suppress degradation, and improve thermal stability. Finally, key challenges and future research directions are outlined to guide the development of robust and sustainable next-generation starting batteries.
figure placeholder
Nicholas B. Watkins et al 2026 J. Electrochem. Soc. 173 112501
Zirconium (Zr) possesses outstanding properties, including exceptional chemical resistance and a high melting point, and is therefore desirable for use in a wide variety of challenging environments, such as in nuclear reactors and the chemical processing industry. To minimize the amount of Zr required for any given application, developing methods for generating metallic Zr coatings is highly advantageous. Owing to Zr’s highly negative reduction potential, electrodeposition in traditional solvents at near ambient temperatures remains challenging and thus not well understood. Due to the extreme conditions required to deposit this metal, extensive work has been conducted in molten salt electrolytes, however the broad applicability of this methodology is limited due to its corrosivity. The primary focus of this article is to present an overview of Zr’s electrochemical behavior and to consolidate the efforts of researchers in exploring electrodeposition techniques for Zr involving aqueous, organic, ionic liquid, deep eutectic, and molten salt solvents. With this information, we highlight trends across solvent systems and opportunities for future research.
figure placeholder
E. Sandrucci et al 2026 J. Electrochem. Soc. 173 100504
This study presents a thorough analysis of machine learning techniques utilized for monitoring the health and predicting the remaining useful life (RUL) of lithium-ion batteries, which is essential for optimizing the performance of electric vehicles (EVs). Given the challenges faced by lithium-ion batteries, such as capacity fading and environmental factors, key indicators like State of Health (SOH) and RUL become vital. The paper reviews several adaptive machine learning methodologies, including Support Vector Regression (SVR), Gaussian Process Regression (GPR), and hybrid neural networks that integrate convolutional neural networks (CNNs) with long short-term memory (LSTM) algorithms. Special emphasis is placed on these models’ effectiveness in addressing the complex nonlinear behaviors associated with battery aging. Moreover, innovative approaches like fuzzy logic systems are examined, showcasing their potential to enhance SOH estimation via adaptable rule-based techniques. The manuscript stresses the necessity of integrating multiple methodologies to enhance predictive accuracy and reliability. By compiling empirical studies, the work aims to elucidate the capabilities of these algorithms, thereby enriching the knowledge base within battery management systems. Ultimately, this research aims to drive advancements in efficient energy storage solutions, which are crucial for the sustainable development of electric mobility.
figure placeholder
Highlights
Machine learning techniques like CNN and DNN significantly enhance the prediction accuracy of battery health by modeling complex relationships.
Gaussian Process Regression (GPR) effectively addresses battery aging issues by flexibly modeling nonlinear relationships and estimating prediction uncertainty.
Condition monitoring utilizes critical parameters such as voltage and temperature to improve state-of-health and remaining useful life assessments.
Machine learning algorithms are essential for predicting battery failure signs and diagnosing degradation trends, thereby enhancing energy storage system reliability.
Various methodologies for estimating state-of-health showcase a mixture of experimental and adaptive techniques aimed at refining battery health evaluation.
Michael M. Thackeray et al 2026 J. Electrochem. Soc. 173 090519
This narrative summarizes, in retrospect, the enormous and quietly-conducted contribution of Dr Johan Coetzer, a South African scientist and entrepreneur, to the discovery, development and implementation of the high temperature sodium—metal chloride “ZEBRA” battery (Na/β-Al2O3, NaAlCl4/MCl2 (M = Ni, Fe)). This research activity was initiated in the mid 1970’s at the Council for Scientific and Industrial Research (CSIR) in South Africa and subsequently developed primarily in partnership with the Atomic Energy Research Establishment (AERE, Harwell, UK), Beta R&D (Derby, UK) and Daimler Benz (Germany), before being transferred to industry worldwide for electric vehicle and energy storage applications, albeit at a relatively low production rate. Coetzer’s wide-ranging, innovative and, at times, unconventional scientific approach also laid the foundation for the discovery and implementation of manganese-based spinel and layered metal oxide cathode materials for the Li-ion battery industry.
figure placeholder
William Lvovich et al 2026 J. Electrochem. Soc. 173 082502
This study investigates aqueous electrolytes for efficient iron electrodeposition directly from Fe3+ at ambient temperature. Concentrated lithium chloride (LiCl) based electrolytes were found to stabilize the Fe3+ in the electrolyte, suppress parasitic hydrogen evolution and suppress redox shuttling of intermediate Fe2+, thus promoting efficient iron electrodeposition. These attributes collectively enabled high Coulombic efficiencies (>75%) at high current densities (>200 mA cm−2). Furthermore, detailed investigation revealed that increasing the LiCl concentration increased the electrolyte viscosity and decreased the Fe2+ out-diffusion rate while also suppressing the activity of water. Sufficiently high LiCl concentrations (>7 M) thus provide a window of current densities in which elevated Coulombic efficiencies can be achieved. Overall, this paper develops an understanding of the effects of electrolyte composition on the competition between Fe electrodeposition, hydrogen co-evolution, and intermediate Fe2+ out-diffusion for efficient Fe metal electrosynthesis.
figure placeholder
Riccardo Candeago et al 2025 J. Electrochem. Soc. 172 113503
Production of volatile fatty acids (VFAs) from biomass fermentation is a promising pathway for sustainable chemical production. A central challenge is cost-effective recovery and fractionation of VFAs from fermentation broths. Redox-mediated electrodialysis (redox-ED) can be an energy efficient technique for VFA separation, replacing water splitting with redox-reactions. However, the low selectivity between VFAs of different lengths (separation factors <3) obtained using commercially-available anion-exchange membranes limits the final product purity for industrial use. Cascaded operation can overcome these material limitations and increase product purity, yet multistage redox-ED can be complex and has not been extensively explored for selective ion separations. Here, we developed a physics-based model of redox-ED using finite elements to simulate the separation of a ternary carboxylic acid feed (propionic, butyric, and hexanoic) and evaluated five different multistage architectures including series, parallel, co-current, and counter-current setups. The single-stage, two-dimensional steady-state model captured the effect of the flow-field, concentration, and potential gradients, yielding reasonable agreement with experimental data. We then investigated the tradeoff between number of stages, product purity, and productivity, and assessed the impact of membrane selectivity. In sum, cascaded redox-ED with reflux simultaneously improved product purity and productivity, while series or equivalent single-stage architectures presented a tradeoff between VFA purity and productivity.
figure placeholder
Maria Kelly et al 2025 J. Electrochem. Soc. 172 046503
Voltammetric measurements of electrochemical CO2 reduction reaction (CO2RR) selectivity on rotating ring disk electrodes (RRDE) are a rapid and sensitive method for quantifying an electrocatalyst’s selectivity, i.e. faradaic efficiency (FE). This method has been applied to polycrystalline Au electrocatalysts where a Au disk electrode catalyzes both the CO2RR and hydrogen evolution reaction while the concentric Au ring electrode selectively senses CO by oxidizing CO back to CO2. Such measurements enabled fundamental mechanistic studies but suffer from poor inter-laboratory reproducibility. This work identifies causes of variability in RRDE selectivity measurements by comparing protocols with different electrochemical methods, reagent purities, and glassware cleaning procedures. We observed FECO decrease by 56% during 5 min chronoamperometry measurements, a phenomenon that is not readily apparent in voltammetric scans due to their dynamic nature. Electroplating of electrolyte impurities onto the disk and ring surfaces were identified as a major contributor to Au deactivation. Additionally, the oxygen reduction reaction may lead to higher disk currents in inadequately purged electrolytes, causing an apparent underestimation of FECO at low overpotentials. Lastly, we propose operational bounds for CO2RR selectivity measurements on Au using the RRDE method and provide suggestions on steps for improving the accuracy of this technique.
figure placeholder
Anar Badalbayli et al 2025 J. Electrochem. Soc. 172 032508
This study explores chloride molten salt electrolysis (CMSE) as a promising route for energy-efficient iron metal (Fe) production. Moderate temperature (500 °C) LiCl-KCl molten salts offer excellent thermodynamic stability, high ionic conductivity and diffusivity, and high solubility for FeCl3, thereby enabling efficient Fe metal extraction at high electrowinning rates. Here, we demonstrate the two essential steps for converting taconite ore into Fe metal. First, Fe2O3 from taconite pellets was selectively leached in HCl yielding a high-purity FeCl3 aqueous solution, while the gangue components settled at the bottom. Then, anhydrous FeCl3 was electrolyzed in a LiCl-KCl eutectic molten salt at 500 °C at high current density (1 A cm−2) and at high Coulombic efficiency (>85%). Analysis of the electrowon Fe deposits revealed dendritic structures with purity of >99 wt%, which could be further improved to nearly 100 wt% through arc re-melting. CMSE offers low specific energy consumption (3.7 kWhr kg−1), competitive with H2-DRI and other electrolytic approaches being pursued globally. Our findings underscore the potential of CMSE as an energy-efficient route for electrosynthesis of Fe metal.
Guoxin Li et al 2025 J. Electrochem. Soc. 172 026501
Cathode-electrolyte interphase (CEI) is critical for inhibiting the cathode degradation to maintain cell life. However, the evolution of the CEI is still unclear due to its complex and slow dynamic process. Here we used scanning electrochemical microscopy (SECM) for in situ investigation of CEI formation process on LiFePO4 cathode. Feedback images and probe scan curves showed a heterogeneous passivation that was gently generated on the LiFePO4 particles during both charging and discharging. Besides, a LiFePO4 composited electrode was also used to investigate the CEI formation to simulate the condition of real battery system. The composited cathode does not show obvious CEI formation within first two cycles. The SECM results between the pristine LiFePO4 particles and the composited LiFePO4 indicated the dynamic accumulation of CEI, which is influenced by the ability to charge transfer kinetics of cathode materials. This approach provided a feasible consideration for the connections between the dynamic evolution of the CEI and changes in charge transfer capability of cathode during cycling.
figure placeholder
Highlights
In-situ investigation of cathode-electrolyte interphase formation.
The evolution of native active material and composite slurry were compared.
The electrochemical activity change upon cathode cycling are analysed in situ.
The influence of the charge transfer capability upon CEI generation is revealed.
Kambe et al
Nanoscale defects within polymer electrolytes disrupt ion transport pathways, resulting in variations in ionic conductivity. Quantifying electrochemical impedance spectroscopy (EIS) signal from connected and disconnected ionic pathways remains challenging without knowing information of all nanoscale transport pathways in polymer electrolytes. In this work, devices with precisely patterned nanoscale defects in ion conduction pathways have been fabricated to quantify the impact of pathway connectivity on ion transport. Each device is designed with a distinct ratio of connected to disconnected pathways. For devices composed exclusively of a single type of ion-conduction pathway, a simple equivalent-circuit model is employed to extract the resistance and capacitance associated with the individual connected or disconnected pathways. This framework is then extended to a unified model that incorporates both connected and disconnected circuit elements, enabling prediction of the EIS spectra for devices containing mixed pathways. The established framework has been applied to successfully fit the experimental data for both dry poly(ethylene oxide) doped with lithium bis(trifluoromethanesulfonyl)imide for Li+ transport and hydrated sulfonated poly(styrene) for H+ transport. This study offers a platform that enables researchers to quantify ionic conductivity contributions from nanoscale connected and disconnected pathways and pave the way towards designing polymer electrolytes with efficient ion transport.
Zhou et al
Silicon (Si)-based materials are prime candidates for superseding conventional graphite in next-generation lithium-ion batteries (LIBs) owing to their exceptional theoretical specific capacities. Nevertheless, their commercial viability is hindered by massive volumetric fluctuations during cycling. Herein, two core–shell composites, Si@acetylene-derived carbon (Si@ACC) and Si@glucose-derived carbon (Si@GCC), were synthesized via chemical vapor deposition (CVD) and hydrothermal–carbonization processes, respectively. The differences between the two carbon shells and their impacts on electrochemical performance are systematically compared. Notably, the carbon layer prepared by the CVD strategy enables a high initial Coulombic efficiency (ICE) of 95.98%. In this core–shell design, the conformal carbon shell buffers expansive stresses while providing rapid electron transport pathways, thereby improving structural integrity and accelerated electrochemical kinetics. The Si@ACC electrode retains 66.33% of its capacity after 300 cycles at 1.0 A g-1. Moreover, a full-cell with a Si@ACC/graphite anode is assembled, delivering an ICE of 83.78% and a capacity retention rate of 66.49% over 200 cycles. Our strategy involves a simple synthesis process to prepare anode material that exhibits improved cycling stability and offers a promising pathway toward realizing the commercial potential of Si anodes for advanced LIBs.
Zulkifli et al
Bisphenol A (BPA) is an endocrine-disrupting compound widely found in plastics and food packaging, posing potential risks to human health and the environment. These concerns highlight the need for sensitive and selective electrochemical detection methods. In this study, chitosan-coated magnetic nanoparticles (MNP-CHT) were developed as a functional sensing material for BPA detection using a magnetic carbon paste electrode (MCPE). Magnetic nanoparticles were synthesized via a co-precipitation method and subsequently modified with chitosan to enhance adsorption and electrochemical activity. The MNP-CHT composite was characterized using FTIR, XRD, and FESEM analyses. Electrochemical studies demonstrated that BPA oxidation on the MNP-CHT MCPE followed a two-electron, two-proton transfer mechanism, as supported by pH and scan rate investigations. Differential pulse voltammetry enabled BPA detection over a linear concentration range of 2–60 μM, with a detection limit of 7.83 nM. The sensor exhibited good reusability for up to five cycles, excellent reproducibility, and stable performance over 21 days. High selectivity and strong anti-interference behavior were achieved in the presence of structurally related BPA analogues, including bisphenol-S, bisphenol-AF, and tetrabromo bisphenol-A. The practical applicability of the proposed sensor was validated through successful BPA determination in tap water, apple juice, and baby teether extracts with satisfactory recovery values.
Naorem et al
A key limitation of scaling electrochemical jet machining (ECJM) to multi-nozzle configurations is the loss of localized machining due to electrolyte interactions and electric-field overlap. This work establishes a quantitative design criterion for maintaining localization in dual-jet ECJM through a combined numerical-experimental approach. A two-dimensional multiphysics model is developed to analyze the coupled behavior of electrolyte flow, electric field distribution, and resulting material removal. The results reveal that nozzle pitch governs both hydraulic-jump interaction and current-density confinement. For a nozzle with inner and outer diameters of 220 µm and 450 µm, respectively, a minimum pitch of 2 mm is identified as the critical threshold to suppress electrolyte overlap and stray corrosion, which is validated experimentally with a prediction error below 6%. In micro-grooving, increasing the traverse speed significantly improves surface finish, reducing RMS roughness (Sq) from 648 nm to 242 nm, but at the cost of reduced depth per pass. To address this trade-off, a feed-assisted multi-pass strategy is introduced, enabling deeper machining while preserving surface quality. Using the optimized conditions, uniform micro-groove arrays with a width of 360 µm, depth of 200 µm, and pitch of 666 µm are fabricated in tungsten with high repeatability and minimal stray corrosion.
Horst et al
The carbon support surface chemistry strongly influences ionomer interactions, water management, and electrochemical performance in polymer electrolyte fuel cell catalyst layers; however, studies that decouple these effects from Pt particle size remain limited. Here, diazonium grafting is applied to commercial 40 wt% Pt/Vulcan and Pt/Ketjenblack catalysts to introduce five aryl functionalities (–NO₂,–NH₂,–CF₃,–COOH,–SO₃H) spanning a wide range of polarities and wettabilities without altering Pt dispersion. Ex situ characterization (x-ray photoelectron spectroscopy, thermogravimetric analysis, N₂ physisorption, contact angle, and dynamic vapor sorption) confirms successful functionalization and shows that grafting on Ketjenblack occurs preferentially at external surfaces, reducing pore accessibility. In 5 cm² single-cell tests, nitrogen-containing functionalities reduce cathode proton transport resistance, increasing current density at 0.7V (1.20 vs 1.01 Acm⁻², I/C=0.6). For hydrophilic and hydrophobic groups, optimal performance depends strongly on the ionomer-to-carbon ratio: hydrophilic functionalities enhance performance at low I/C (0.1) but become detrimental at higher I/C (0.6) due to increased water retention and unfavorable ionomer interactions. These effects are amplified for porous Ketjenblack, where pore blockage offsets gains from surface modification. These findings establish that surface functionalization and support porosity cannot be optimized independently, and suggest that next-generation mesoporous carbon supports could incorporate pore architectures compatible with surface modification.
Trending on Altmetric
Yohei Nagatomo et al 2026 J. Electrochem. Soc. 173 114508
A unified theoretical framework is proposed for describing the exchange current density of electrode reactions in solid oxide cells, covering charge transfer, dissociative adsorption, and surface transport as possible rate-determining steps. By combining kinetic processes with equilibrium relations among dissociative adsorption equilibria, the apparent dependencies of exchange current density on gas partial pressures are systematically derived, together with general forms that also accommodate temperature dependence. The apparent activation energy can be expressed as the sum of the intrinsic barrier of the rate-determining step and the standard enthalpies of quasi-equilibrated reactions, weighted by surface coverage. Apparent reaction orders are likewise formulated as the combination of kinetic and thermodynamic contributions, revealing fundamental constraints on their accessible values. As a concrete example, analysis of the H2/H2O system demonstrates that the possible combinations of reaction orders fall within a parallelogram-shaped region defined by ideal Langmuir adsorption equilibria. This result illustrates how this framework not only explains empirical power-law relations but also helps clarify why distinct elementary mechanisms can yield indistinguishable pressure dependences. This approach provides a consistent basis for interpreting electrode kinetics and offers a general tool for linking microscopic reaction models with macroscopic experimental observables in solid oxide cells.
figure placeholder
Highlights
Explains gas-pressure dependencies of exchange current in solid oxide cells.
Dependencies combine kinetic effects with thermodynamics via surface coverage.
H2/H2O reaction places apparent pressure exponents inside a parallelogram region.
Highlights non-uniqueness of pressure exponents across elementary mechanisms.
Rajan Maurya et al 2026 J. Electrochem. Soc. 173 114507
The effect of pre-wetting on the catalyst utilization and durability of high-temperature proton exchange membrane fuel cell (HT-PEMFC) gas diffusion electrodes (GDEs) was investigated in a three-electrode configuration. Pre-wetting protocols included treatment in ethanol-water mixture (0, 10, 50, and 100% v/v ethanol), ethanol-water vapor, and concentrated orthophosphoric acid (14.85 M) at 150 °C. All pre-wetting approaches significantly enhanced electrode wettability by reducing hydrophobicity and surface tension, thereby improving initial catalyst utilization. However, accelerated stress test (AST) revealed a trade-off between utilization and durability, with wetted electrodes exhibiting lower electrochemically active surface area and Pt retention than untreated electrodes. Changes in Pt crystallite and particle sizes during the AST were estimated through X-ray diffraction analysis and transmission electron microscopy. All three wetting procedures have similar effects on catalyst utilization and durability without compromising the structural integrity. Single-cell measurements showed that while pre-wetting is effective for rapid screening and improving the initial performance, its benefits are not directly transferable to practical operation without further optimization. These findings highlight the trade-off between catalyst utilization and durability in HT-PEMFC GDEs.
figure placeholder
Highlights
Proper wetting of HT-PEMFC electrode in aqueous electrolytes is challenging
Methods to improve wettability (alcoholic solvent/temperature) are suggested as protocols
Initial activity and durability of the electrodes depend strongly on the wetting method
Ridge M. Bachman et al 2026 J. Electrochem. Soc. 173 110514
The transition to new energy resources is driving the need for efficient energy storage. Redox flow batteries (RFBs) are a promising solution due to their flexible design and safer materials, particularly for long-duration storage applications. Despite these advantages, RFB technologies still have considerable room for growth in terms of utilizing all of their available energy storage density. Herein, we assess RFB energy storage density utilization trends for multiple chemistries by accounting for their unique thermodynamic limits, energy efficiencies, and charge capacity constraints. Of the RFBs chemistries analyzed, most still use less than 60% of their theoretical energy storage capacity. The conventional all-vanadium RFB was the closest to its thermodynamic limit available. Moreover, the volumetric footprint and electrolyte cost analyses demonstrate the real-world implications of these inefficiencies. This analysis further indicates that future studies would benefit from clearer reporting of two key quantities: discharge efficiency, which is more directly tied to practically recoverable energy density than round-trip energy efficiency, and state-of-charge or capacity-utilization limits, which govern access to theoretical storage capacity. More consistent reporting of these factors would improve the accuracy and comparability of energy storage utilization analyses across RFB chemistries.
figure placeholder
Highlights
Energy storage density values from energy efficiency and capacity constraints
Redox flow batteries use less than 60% of their energy storage density limit
Inefficiencies in redox flow batteries increase their volumetric footprint
Cost effective electrolytes make up for underutilized energy storage density
Yu Kambe et al 2026 J. Electrochem. Soc.
Nanoscale defects within polymer electrolytes disrupt ion transport pathways, resulting in variations in ionic conductivity. Quantifying electrochemical impedance spectroscopy (EIS) signal from connected and disconnected ionic pathways remains challenging without knowing information of all nanoscale transport pathways in polymer electrolytes. In this work, devices with precisely patterned nanoscale defects in ion conduction pathways have been fabricated to quantify the impact of pathway connectivity on ion transport. Each device is designed with a distinct ratio of connected to disconnected pathways. For devices composed exclusively of a single type of ion-conduction pathway, a simple equivalent-circuit model is employed to extract the resistance and capacitance associated with the individual connected or disconnected pathways. This framework is then extended to a unified model that incorporates both connected and disconnected circuit elements, enabling prediction of the EIS spectra for devices containing mixed pathways. The established framework has been applied to successfully fit the experimental data for both dry poly(ethylene oxide) doped with lithium bis(trifluoromethanesulfonyl)imide for Li+ transport and hydrated sulfonated poly(styrene) for H+ transport. This study offers a platform that enables researchers to quantify ionic conductivity contributions from nanoscale connected and disconnected pathways and pave the way towards designing polymer electrolytes with efficient ion transport.
Matthias F. Ernst et al 2026 J. Electrochem. Soc. 173 114501
In this work, three commercial, titanium fiber- or powder-based porous transport layers (PTLs) for polymer electrolyte membrane water electrolysis (PEMWE) are characterized in terms of pore size, porosity, surface roughness, and in-plane permeability. The latter is quantified using an in-house designed setup, revealing a nearly 10-fold higher permeability for fiber compared to powder-based PTLs. PEMWE polarization curves and high-frequency resistances are recorded with 5 cm2 single-cells with serpentine flow fields (FFs), comparing the performance of the three anode PTLs in dependence of the anode FF land width (ranging from 0.7–6.2 mm; with a constant channel width and depth of 1 mm). A detailed voltage loss analysis reveals the absence of significant mass transport resistances at the anode side at low to moderate current densities, even when using the anode FF with the largest land width in combination with the PTL with the lowest permeability. In contrast, at high current densities (>2 A cm−2), the combination of large anode FF land width and low-permeability PTL leads to significant mass transport related performance losses, accompanied by a local dry-out of the membrane and a reduced MEA utilization, which can be rationalized by a simple model.
figure placeholder
Takahiro Suzuki et al 2026 J. Electrochem. Soc. 173 114506
This study examined the correlation between particle agglomeration and the rheological properties of electrode slurries composed of porous electrode materials for polymer electrolyte fuel cells and presented two different agglomeration mechanisms in electrode slurries. Electrode slurries are used to fabricate porous electrodes. The state of electrode slurries is influenced by the materials, dispersion medium, and dispersion conditions, and is believed to affect the resulting porous electrode structure. However, the dispersion state of the materials within the electrode slurries remains inadequately understood. This study identified peculiar rheological behavior in the rheological measurements of electrode slurries, which arises depending on the composition ratio of the materials and dispersion conditions. The formation of two distinct particle agglomerate structures that differed in scale and formation mechanisms within the electrode slurries and their correlation with the rheological properties were revealed based on experimental approaches, including centrifugal classification, X-ray radiography, and rheo-small-angle X-ray scattering (rheo-SAXS).
figure placeholder
Rens J. Horst and Antoni Forner-Cuenca 2026 J. Electrochem. Soc.
The carbon support surface chemistry strongly influences ionomer interactions, water management, and electrochemical performance in polymer electrolyte fuel cell catalyst layers; however, studies that decouple these effects from Pt particle size remain limited. Here, diazonium grafting is applied to commercial 40 wt% Pt/Vulcan and Pt/Ketjenblack catalysts to introduce five aryl functionalities (–NO₂,–NH₂,–CF₃,–COOH,–SO₃H) spanning a wide range of polarities and wettabilities without altering Pt dispersion. Ex situ characterization (x-ray photoelectron spectroscopy, thermogravimetric analysis, N₂ physisorption, contact angle, and dynamic vapor sorption) confirms successful functionalization and shows that grafting on Ketjenblack occurs preferentially at external surfaces, reducing pore accessibility. In 5 cm² single-cell tests, nitrogen-containing functionalities reduce cathode proton transport resistance, increasing current density at 0.7V (1.20 vs 1.01 Acm⁻², I/C=0.6). For hydrophilic and hydrophobic groups, optimal performance depends strongly on the ionomer-to-carbon ratio: hydrophilic functionalities enhance performance at low I/C (0.1) but become detrimental at higher I/C (0.6) due to increased water retention and unfavorable ionomer interactions. These effects are amplified for porous Ketjenblack, where pore blockage offsets gains from surface modification. These findings establish that surface functionalization and support porosity cannot be optimized independently, and suggest that next-generation mesoporous carbon supports could incorporate pore architectures compatible with surface modification.
Haruki Amako et al 2026 J. Electrochem. Soc.
Molten salt electrolysis in high-temperature melts containing carbonate ions is a promising approach for oxygen extraction from CO2 as a feedstock, but CO₂ evolution at the anode often reduces process efficiency. This study proposes a dual-compartment molten salt electrolysis system using a yttria-stabilized zirconia (YSZ) membrane combined with a LaCoO₃ anode to suppress CO₂ evolution in the molten salts. Electrolysis was conducted in LiCl–CaCl₂ melts containing CaO and CaCO₃ at 873 K. O2⁻ ions formed at the cathode by the reduction of carbonate ions were transported through the YSZ membrane to the anode compartment, where oxygen evolution occurred on the LaCoO₃ electrode with high current efficiency. Compared with conventional single-compartment electrolysis, the introduction of the YSZ membrane effectively suppressed CO₂ evolution at the anode. Post-electrolysis analyses confirmed that the YSZ membrane retained its crystal structure without noticeable degradation. Analysis based on the Nernst–Planck equation indicated that migration driven by the electric field plays a dominant role in O2⁻ ion transport through the membrane. These results demonstrate that combining a YSZ membrane with a LaCoO₃ anode enables effective separation of electrochemical reactions and provides a promising approach for controlling anodic reactions in molten salt electrolysis systems.
Michelle L. Lehmann et al 2026 J. Electrochem. Soc. 173 110512
Sodium-sulfur (Na-S) batteries hold promise as high-energy-density batteries for long-duration energy storage due to their high open circuit voltage and Earth-abundant active materials. However, low utilization of the sulfur active material limits the achievable specific capacity, especially when a liquid polysulfide catholyte is utilized. High-surface-area carbon materials demonstrate promise to increase sulfur utilization in a Na-S battery. To make the carbon host material applicable for a flow battery application, a binder is required to bind the carbon materials together and to a porous substrate. Here, we utilize a composite carbon paper electrode coated by polymer,carbon nanofibers (CNFs) and carbon black (CB). By varying the mass ratios of CNF to CB, we found that a 70/20/10 mass ratio of CNF/CB/binder provided the highest capacity (260 mAh g−1 compared to the baseline of 156 mAh g−1) and stable battery performance over 100 cycles. Analysis of the composite electrode and full cells reveals that the amount of CB in the composite electrode influences binder distribution, cell resistance, and full cell performance.
figure placeholder
I. Nesterova et al 2026 J. Electrochem. Soc. 173 110511
The increasing demand for sustainable and safe energy storage technologies has accelerated research into aqueous sodium-ion batteries (SIBs). This study investigates the electrochemical performance and degradation mechanisms of Na2FeP2O7/C (NFP) as a cathode material in aqueous SIBs. We evaluate NFP/C using cyclic voltammetry, galvanostatic charge-discharge cycling, X-ray diffraction, SEM, and ICP-MS, in both half-cell and symmetrical full-cell configurations. NFP/C exhibits an initial capacity of up to 83 mAh g−1 in aqueous half-cells due to continuous Fe release into the electrolyte and the formation of a surface layer that progressively impedes Na⁺ insertion. Symmetrical full-cells demonstrate improved stability, retaining approximately 50 mAh g−1 over 100+ cycles with near-100% Coulombic efficiency. Structural analysis confirms that the bulk pyrophosphate framework is preserved during both chemical immersion and electrochemical cycling, although surface-level reactivity with the electrolyte is detected even at open circuit through the formation of a minor secondary crystalline phase. These findings suggest the potential of NFP/C for aqueous SIBs if strategies to suppress surface degradation can be developed.
figure placeholder
Wolfgang Schmickler and Sergio Trasatti 2006 J. Electrochem. Soc. 153 L31
We compare the adsorption energies calculated by Nørskov et al. with data derived from experimental values; except for Ni and Co, which absorb hydrogen strongly, there is a linear relation. We discuss the model proposed by these authors in the light of extensive previous work and of experimental data and find it overly simplistic.
J. K. Nørskov et al 2005 J. Electrochem. Soc. 152 J23
A density functional theory database of hydrogen chemisorption energies on close packed surfaces of a number of transition and noble metals is presented. The bond energies are used to understand the trends in the exchange current for hydrogen evolution. A volcano curve is obtained when measured exchange currents are plotted as a function of the calculated hydrogen adsorption energies and a simple kinetic model is developed to understand the origin of the volcano. The volcano curve is also consistent with Pt being the most efficient electrocatalyst for hydrogen evolution. © 2005 The Electrochemical Society. All rights reserved.
E. Peled and S. Menkin 2017 J. Electrochem. Soc. 164 A1703
The Solid-Electrolyte-Interphase (SEI) model for non-aqueous alkali-metal batteries constitutes a paradigm change in the understanding of lithium batteries and has thus enabled the development of safer, durable, higher-power and lower-cost lithium batteries for portable and EV applications. Prior to the publication of the SEI model (1979), researchers used the Butler-Volmer equation, in which a direct electron transfer from the electrode to lithium cations in the solution is assumed. The SEI model proved that this is a mistaken concept and that, in practice, the transfer of electrons from the electrode to the solution in a lithium battery, must be prevented, since it will result in fast self-discharge of the active materials and poor battery performance. This model provides [E. Peled, in “Lithium Batteries,” J.P. Gabano (ed), Academic Press, (1983), E. Peled, J. Electrochem. Soc., 126, 2047 (1979).] new equations for: electrode kinetics (io and b), anode corrosion, SEI resistivity and growth rate and irreversible capacity loss of lithium-ion batteries. This model became a cornerstone in the science and technology of lithium batteries. This paper reviews the past, present and the future of SEI batteries.
Roland Jung et al 2017 J. Electrochem. Soc. 164 A1361
Layered LiNixMnyCozO2 (NMC) is a widely used class of cathode materials with LiNi1/3Mn1/3Co1/3O2 (NMC111) being the most common representative. However, Ni-rich NMCs are more and more in the focus of current research due to their higher specific capacity and energy. In this work we will compare LiNi1/3Mn1/3Co1/3O2 (NMC111), LiNi0.6Mn0.2Co0.2O2 (NMC622), and LiNi0.8Mn0.1Co0.1O2 (NMC811) with respect to their cycling stability in NMC-graphite full-cells at different end-of-charge potentials. It will be shown that stable cycling is possible up to 4.4 V for NMC111 and NMC622 and only up to 4.0 V for NMC811. At higher potentials, significant capacity fading was observed, which was traced back to an increase in the polarization of the NMC electrode, contrary to the nearly constant polarization of the graphite electrode. Furthermore, we show that the increase in the polarization occurs when the NMC materials are cycled up to a high-voltage feature in the dq/dV plot, which occurs at ∼4.7 V vs. Li/Li+ for NMC111 and NMC622 and at ∼4.3 V vs. Li/Li+ for NMC811. For the latter material, this feature corresponds to the H2 → H3 phase transition. Contrary to the common understanding that the electrochemical oxidation of carbonate electrolytes causes the CO2 and CO evolution at potentials above 4.7 V vs. Li/Li+, we believe that the observed CO2 and CO are mainly due to the chemical reaction of reactive lattice oxygen with the electrolyte. This hypothesis is based on gas analysis using On-line Electrochemical Mass Spectrometry (OEMS), by which we prove that all three materials release oxygen from the particle surface and that the oxygen evolution coincides with the onset of CO2 and CO evolution. Interestingly, the onsets of oxygen evolution for the different NMCs correlate well with the high-voltage redox feature at ∼4.7 V vs. Li/Li+ for NMC111 and NMC622 as well as at ∼4.3 V vs. Li/Li+ for NMC811. To support this hypothesis, we show that no CO2 or CO is evolved for the LiNi0.43Mn1.57O4 (LNMO) spinel up to 5 V vs. Li/Li+, consistent with the absence of oxygen release. Lastly, we demonstrate by the use of 13C labeled conductive carbon that it is the electrolyte rather than the conductive carbon which is oxidized by the released lattice oxygen. Taking these findings into consideration, a mechanism is proposed for the reaction of released lattice oxygen with ethylene carbonate yielding CO2, CO, and H2O.
D. A. Stevens and J. R. Dahn 2000 J. Electrochem. Soc. 147 1271
Electrochemical techniques have been used to study the reversible insertion of sodium into hard‐carbon host structures at room temperature. In this paper we compare these results with those for lithium insertion in the same materials and demonstrate the presence of similar alkali metal insertion mechanisms in both cases. Despite the gravimetric capacities being lower for sodium than lithium insertion, we have achieved a reversible sodium capacity of 300 mAh/g, close to that for lithium insertion in graphitic carbon anode materials. Such materials may therefore be useful as anodes in rechargeable sodium‐ion batteries. © 2000 The Electrochemical Society. All rights reserved.
Todd M. Bandhauer et al 2011 J. Electrochem. Soc. 158 R1
Lithium-ion batteries are well-suited for fully electric and hybrid electric vehicles due to their high specific energy and energy density relative to other rechargeable cell chemistries. However, these batteries have not been widely deployed commercially in these vehicles yet due to safety, cost, and poor low temperature performance, which are all challenges related to battery thermal management. In this paper, a critical review of the available literature on the major thermal issues for lithium-ion batteries is presented. Specific attention is paid to the effects of temperature and thermal management on capacity/power fade, thermal runaway, and pack electrical imbalance and to the performance of lithium-ion cells at cold temperatures. Furthermore, insights gained from previous experimental and modeling investigations are elucidated. These include the need for more accurate heat generation measurements, improved modeling of the heat generation rate, and clarity in the relative magnitudes of the various thermal effects observed at high charge and discharge rates seen in electric vehicle applications. From an analysis of the literature, the requirements for lithium-ion thermal management systems for optimal performance in these applications are suggested, and it is clear that no existing thermal management strategy or technology meets all these requirements.
D. A. Stevens and J. R. Dahn 2001 J. Electrochem. Soc. 148 A803
In this paper, we compare the interactions of lithium and sodium with a range of carbon materials in electrochemical cells. Through wide angle in situ X-ray scattering studies, we demonstrate that both lithium and sodium can be inserted into the interlayer space in disordered carbon materials. This insertion process is accompanied by an increase in the interlayer spacing in these materials. Small-angle in situ scattering studies are presented to clearly show the insertion of lithium and sodium into nanopores within disordered hard carbons. We also show that very little, if any, sodium can be inserted into graphitic materials in contrast to the large capacity seen for lithium insertion. © 2001 The Electrochemical Society. All rights reserved.
Charles Monroe and John Newman 2005 J. Electrochem. Soc. 152 A396
Past theories of electrode stability assume that the surface tension resists the amplification of surface roughness at cathodes and show that instability at lithium/liquid interfaces cannot be prevented by surface forces alone [Electrochim. Acta, 40, 599 (1995)]. This work treats interfacial stability in lithium/polymer systems where the electrolyte is solid. Linear elasticity theory is employed to compute the additional effect of bulk mechanical forces on electrode stability. The lithium and polymer are treated as Hookean elastic materials, characterized by their shear moduli and Poisson’s ratios. Two-dimensional displacement distributions that satisfy force balances across a periodically deforming interface are derived; these allow computation of the stress and surface-tension forces. The incorporation of elastic effects into a kinetic model demonstrates regimes of electrolyte mechanical properties where amplification of surface roughness can be inhibited. For a polymer material with Poisson’s ratio similar to poly(ethylene oxide), interfacial roughening is mechanically suppressed when the separator shear modulus is about twice that of lithium. © 2005 The Electrochemical Society. All rights reserved.
Wenchao Sheng et al 2010 J. Electrochem. Soc. 157 B1529
The kinetics of the hydrogen oxidation reaction (HOR) and hydrogen evolution reaction (HER) on polycrystalline platinum [Pt(pc)] and high surface area carbon-supported platinum nanoparticles (Pt/C) were studied in 0.1 M KOH using rotating disk electrode (RDE) measurements. After corrections of noncompensated solution resistance from ac impedance spectroscopy and of hydrogen mass transport in the HOR branch, the kinetic current densities were fitted to the Butler–Volmer equation using a transfer coefficient of 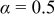 , from which HOR/HER exchange current densities on Pt(pc) and Pt/C were obtained, and the HOR/HER mechanisms in alkaline solution were discussed. Unlike the HOR/HER rates on Pt electrodes in alkaline solution, the HOR/HER rates on a Pt electrode in 0.1 M
, from which HOR/HER exchange current densities on Pt(pc) and Pt/C were obtained, and the HOR/HER mechanisms in alkaline solution were discussed. Unlike the HOR/HER rates on Pt electrodes in alkaline solution, the HOR/HER rates on a Pt electrode in 0.1 M 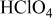 were limited entirely by hydrogen diffusion, which renders the quantification of the HOR/HER kinetics impossible by conventional RDE measurements. The simulation of the hydrogen anode performance based on the specific exchange current densities of the HOR/HER at
were limited entirely by hydrogen diffusion, which renders the quantification of the HOR/HER kinetics impossible by conventional RDE measurements. The simulation of the hydrogen anode performance based on the specific exchange current densities of the HOR/HER at 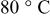 illustrates that in addition to the oxygen reduction reaction cell voltage loss on the cathode, the slow HOR kinetics are projected to cause significant anode potential losses in alkaline fuel cells for low platinum loadings (
illustrates that in addition to the oxygen reduction reaction cell voltage loss on the cathode, the slow HOR kinetics are projected to cause significant anode potential losses in alkaline fuel cells for low platinum loadings (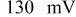 at
at 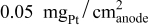 and
and 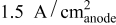 ), contrary to what is reported for proton exchange membrane fuel cells.
), contrary to what is reported for proton exchange membrane fuel cells.
George E. Blomgren 2017 J. Electrochem. Soc. 164 A5019
This year, the battery industry celebrates the 25th anniversary of the introduction of the lithium ion rechargeable battery by Sony Corporation. The discovery of the system dates back to earlier work by Asahi Kasei in Japan, which used a combination of lower temperature carbons for the negative electrode to prevent solvent degradation and lithium cobalt dioxide modified somewhat from Goodenough's earlier work. The development by Sony was carried out within a few years by bringing together technology in film coating from their magnetic tape division and electrochemical technology from their battery division. The past 25 years has shown rapid growth in the sales and in the benefits of lithium ion in comparison to all the earlier rechargeable battery systems. Recent work on new materials shows that there is a good likelihood that the lithium ion battery will continue to improve in cost, energy, safety and power capability and will be a formidable competitor for some years to come.
Journal links
ECS Publications
- Journal of the Electrochemical Society
- ECS Journal of Solid State Science and Technology
- ECS Advances
- ECS Sensors Plus
- ECS Transactions
- The Electrochemical Society Interface
- ECS Meeting Abstracts
- ECS Electrochemistry Letters
- ECS Solid State Letters
- Electrochemical and Solid State Letters
- ECS Proceedings Volumes
Journal information
- 1902-present
Journal of The Electrochemical Society
doi: 10.1149/issn.1945-7111
Online ISSN: 1945-7111
Print ISSN: 0013-4651