The maximum energy that lithium-ion batteries can store decreases as they are used because of various irreversible degradation mechanisms. Many models of degradation have been proposed in the literature, sometimes with a small experimental data set for validation. However, a comprehensive comparison between different model predictions is lacking, making it difficult to select modelling approaches which can explain the degradation trends actually observed from data. Here, various degradation models from literature are implemented within a single particle model framework and their behavior is compared. It is shown that many different models can be fitted to a small experimental data set. The interactions between different models are simulated, showing how some of the models accelerate degradation in other models, altering the overall degradation trend. The effects of operating conditions on the various degradation models is simulated. This identifies which models are enhanced by which operating conditions and might therefore explain specific degradation trends observed in data. Finally, it is shown how a combination of different models is needed to capture different degradation trends observed in a large experimental data set. Vice versa, only a large data set enables to properly select the models which best explain the observed degradation.

The Electrochemical Society (ECS) was founded in 1902 to advance the theory and practice at the forefront of electrochemical and solid state science and technology, and allied subjects.
ISSN: 1945-7111
JES is the flagship journal of The Electrochemical Society. Published continuously from 1902 to the present, JES remains one of the most highly-cited journals in electrochemistry and solid-state science and technology.
Jorn M. Reniers et al 2019 J. Electrochem. Soc. 166 A3189
Eniko S. Zsoldos et al 2024 J. Electrochem. Soc. 171 080527
Lithium iron phosphate (LFP) battery cells are ubiquitous in electric vehicles and stationary energy storage because they are cheap and have a long lifetime. This work compares LFP/graphite pouch cells undergoing charge-discharge cycles over five state of charge (SOC) windows (0%–25%, 0%–60%, 0%–80%, 0%–100%, and 75%–100%). Cycling LFP cells across a lower average SOC results in less capacity fade than cycling across a higher average SOC, regardless of depth of discharge. The primary capacity fade mechanism is lithium inventory loss due to: lithiated graphite reactivity with electrolyte, which increases incrementally with SOC, and lithium alkoxide species causing iron dissolution and deposition on the negative electrode at high SOC which further accelerates lithium inventory loss. Our results show that even low voltage LFP systems (3.65 V) have a tradeoff between average SOC and lifetime. Operating LFP cells at lower average SOC can extend their lifetime substantially in both EV and grid storage applications.
Peter Keil et al 2016 J. Electrochem. Soc. 163 A1872
In this study, the calendar aging of lithium-ion batteries is investigated at different temperatures for 16 states of charge (SoCs) from 0 to 100%. Three types of 18650 lithium-ion cells, containing different cathode materials, have been examined. Our study demonstrates that calendar aging does not increase steadily with the SoC. Instead, plateau regions, covering SoC intervals of more than 20%–30% of the cell capacity, are observed wherein the capacity fade is similar. Differential voltage analyses confirm that the capacity fade is mainly caused by a shift in the electrode balancing. Furthermore, our study reveals the high impact of the graphite electrode on calendar aging. Lower anode potentials, which aggravate electrolyte reduction and thus promote solid electrolyte interphase growth, have been identified as the main driver of capacity fade during storage. In the high SoC regime where the graphite anode is lithiated more than 50%, the low anode potential accelerates the loss of cyclable lithium, which in turn distorts the electrode balancing. Aging mechanisms induced by high cell potential, such as electrolyte oxidation or transition-metal dissolution, seem to play only a minor role. To maximize battery life, high storage SoCs corresponding to low anode potential should be avoided.
Chang-Hui Chen et al 2020 J. Electrochem. Soc. 167 080534
Presented here, is an extensive 35 parameter experimental data set of a cylindrical 21700 commercial cell (LGM50), for an electrochemical pseudo-two-dimensional (P2D) model. The experimental methodologies for tear-down and subsequent chemical, physical, electrochemical kinetics and thermodynamic analysis, and their accuracy and validity are discussed. Chemical analysis of the LGM50 cell shows that it is comprised of a NMC 811 positive electrode and bi-component Graphite-SiOx negative electrode. The thermodynamic open circuit voltages (OCV) and lithium stoichiometry in the electrode are obtained using galvanostatic intermittent titration technique (GITT) in half cell and three-electrode full cell configurations. The activation energy and exchange current coefficient through electrochemical impedance spectroscopy (EIS) measurements. Apparent diffusion coefficients are estimated using the Sand equation on the voltage transient during the current pulse; an expansion factor was applied to the bi-component negative electrode data to reflect the average change in effective surface area during lithiation. The 35 parameters are applied within a P2D model to show the fit to experimental validation LGM50 cell discharge and relaxation voltage profiles at room temperature. The accuracy and validity of the processes and the techniques in the determination of these parameters are discussed, including opportunities for further modelling and data analysis improvements.
Anu Adamson et al 2026 J. Electrochem. Soc. 173 060508
This study presents post-mortem analyses of NMC/graphite pouch cells, which exhibit exceptional capacity retention after up to 8 years or 22,000 charge-discharge cycles. Various NMC/graphite cells with different positive electrode compositions and particle morphologies were examined under a variety of cycling conditions, temperatures, C-rates, and upper cut-off voltages. The findings demonstrate that even after extended periods of continuous cycling, the NMC positive electrodes can maintain the integrity of their crystal structure with minimal transition metal dissolution. Additionally, there is minimal degradation of the electrolyte, and the artificial graphite (AG) negative electrode materials only show negligible changes. Ultimately, lithium loss at the negative electrode is the only notable aging mode in these cells. Furthermore, with optimized electrolytes and high-quality artificial graphite, this parasitic reaction occurs at a low rate. As a result, lithium-ion cells can achieve remarkably long lifetimes. For example, a single-crystal NMC532/AG cell studied in this research retained 80% of its capacity, positive electrode crystal structure, and morphology, with minimal changes to the electrolyte after 6.8 years of continuous charge-discharge cycling.
figure placeholder
A. K. Padhi et al 1997 J. Electrochem. Soc. 144 1188
Reversible extraction of lithium from 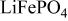 (triphylite) and insertion of lithium into
(triphylite) and insertion of lithium into 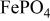 at 3.5 V vs. lithium at 0.05 mA/cm2 shows this material to be an excellent candidate for the cathode of a low‐power, rechargeable lithium battery that is inexpensive, nontoxic, and environmentally benign. Electrochemical extraction was limited to ∼0.6 Li/formula unit; but even with this restriction the specific capacity is 100 to 110 mAh/g. Complete extraction of lithium was performed chemically; it gave a new phase,
at 3.5 V vs. lithium at 0.05 mA/cm2 shows this material to be an excellent candidate for the cathode of a low‐power, rechargeable lithium battery that is inexpensive, nontoxic, and environmentally benign. Electrochemical extraction was limited to ∼0.6 Li/formula unit; but even with this restriction the specific capacity is 100 to 110 mAh/g. Complete extraction of lithium was performed chemically; it gave a new phase, 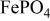 , isostructural with heterosite,
, isostructural with heterosite,  . The
. The 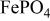 framework of the ordered olivine
framework of the ordered olivine 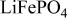 is retained with minor displacive adjustments. Nevertheless the insertion/extraction reaction proceeds via a two‐phase process, and a reversible loss in capacity with increasing current density appears to be associated with a diffusion‐limited transfer of lithium across the two‐phase interface. Electrochemical extraction of lithium from isostructural
is retained with minor displacive adjustments. Nevertheless the insertion/extraction reaction proceeds via a two‐phase process, and a reversible loss in capacity with increasing current density appears to be associated with a diffusion‐limited transfer of lithium across the two‐phase interface. Electrochemical extraction of lithium from isostructural 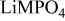 (M = Mn, Co, or Ni) with an
(M = Mn, Co, or Ni) with an 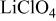 electrolyte was not possible; but successful extraction of lithium from
electrolyte was not possible; but successful extraction of lithium from 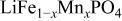 was accomplished with maximum oxidation of the
was accomplished with maximum oxidation of the 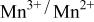 occurring at x = 0.5. The
occurring at x = 0.5. The 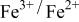 couple was oxidized first at 3.5 V followed by oxidation of the
couple was oxidized first at 3.5 V followed by oxidation of the 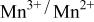 couple at 4.1 V vs. lithium. The
couple at 4.1 V vs. lithium. The 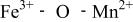 interactions appear to destabilize the
interactions appear to destabilize the 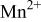 level and stabilize the
level and stabilize the 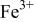 level so as to make the
level so as to make the 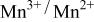 energy accessible.
energy accessible.
Manuel Ank et al 2023 J. Electrochem. Soc. 170 120536
Battery research depends upon up-to-date information on the cell characteristics found in current electric vehicles, which is exacerbated by the deployment of novel formats and architectures. This necessitates open access to cell characterization data. Therefore, this study examines the architecture and performance of first-generation Tesla 4680 cells in detail, both by electrical characterization and thermal investigations at cell-level and by disassembling one cell down to the material level including a three-electrode analysis. The cell teardown reveals the complex cell architecture with electrode disks of hexagonal symmetry as well as an electrode winding consisting of a double-sided and homogeneously coated cathode and anode, two separators and no mandrel. A solvent-free anode fabrication and coating process can be derived. Energy-dispersive X-ray spectroscopy as well as differential voltage, incremental capacity and three-electrode analysis confirm a NMC811 cathode and a pure graphite anode without silicon. On cell-level, energy densities of 622.4 Wh/L and 232.5 Wh/kg were determined while characteristic state-of-charge dependencies regarding resistance and impedance behavior are revealed using hybrid pulse power characterization and electrochemical impedance spectroscopy. A comparatively high surface temperature of ∼70 °C is observed when charging at 2C without active cooling. All measurement data of this characterization study are provided as open source.
Yuliya Preger et al 2020 J. Electrochem. Soc. 167 120532
Energy storage systems with Li-ion batteries are increasingly deployed to maintain a robust and resilient grid and facilitate the integration of renewable energy resources. However, appropriate selection of cells for different applications is difficult due to limited public data comparing the most commonly used off-the-shelf Li-ion chemistries under the same operating conditions. This article details a multi-year cycling study of commercial LiFePO4 (LFP), LiNixCoyAl1−x−yO2 (NCA), and LiNixMnyCo1−x−yO2 (NMC) cells, varying the discharge rate, depth of discharge (DOD), and environment temperature. The capacity and discharge energy retention, as well as the round-trip efficiency, were compared. Even when operated within manufacturer specifications, the range of cycling conditions had a profound effect on cell degradation, with time to reach 80% capacity varying by thousands of hours and cycle counts among cells of each chemistry. The degradation of cells in this study was compared to that of similar cells in previous studies to identify universal trends and to provide a standard deviation for performance. All cycling files have been made publicly available at batteryarchive.org, a recently developed repository for visualization and comparison of battery data, to facilitate future experimental and modeling efforts.
E. Peled and S. Menkin 2017 J. Electrochem. Soc. 164 A1703
The Solid-Electrolyte-Interphase (SEI) model for non-aqueous alkali-metal batteries constitutes a paradigm change in the understanding of lithium batteries and has thus enabled the development of safer, durable, higher-power and lower-cost lithium batteries for portable and EV applications. Prior to the publication of the SEI model (1979), researchers used the Butler-Volmer equation, in which a direct electron transfer from the electrode to lithium cations in the solution is assumed. The SEI model proved that this is a mistaken concept and that, in practice, the transfer of electrons from the electrode to the solution in a lithium battery, must be prevented, since it will result in fast self-discharge of the active materials and poor battery performance. This model provides [E. Peled, in “Lithium Batteries,” J.P. Gabano (ed), Academic Press, (1983), E. Peled, J. Electrochem. Soc., 126, 2047 (1979).] new equations for: electrode kinetics (io and b), anode corrosion, SEI resistivity and growth rate and irreversible capacity loss of lithium-ion batteries. This model became a cornerstone in the science and technology of lithium batteries. This paper reviews the past, present and the future of SEI batteries.
D. Bernardi et al 1985 J. Electrochem. Soc. 132 5
A general energy balance for battery systems has been developed. This equation is useful for estimating cell thermal characteristics. Reliable predictions of cell temperature and heat‐generation rate are required for the design and thermal management of battery systems. The temperature of a cell changes as a result of electrochemical reactions, phase changes, mixing effects, and joule heating. The equation developed incorporates these effects in a complete and general manner. Simplifications and special cases are discussed. The results of applying the energy balance to a mathematical model of the  cell discharged through two different reaction mechanisms are given as examples. The examples illustrate how the energy equation may be applied to a specific system to examine the relative contributions corresponding to the terms in the equation. The examples show that the processes involved in cell heat generation may be complex and that the application of a sufficiently general energy equation is advantageous.
cell discharged through two different reaction mechanisms are given as examples. The examples illustrate how the energy equation may be applied to a specific system to examine the relative contributions corresponding to the terms in the equation. The examples show that the processes involved in cell heat generation may be complex and that the application of a sufficiently general energy equation is advantageous.
Marcel Rogge et al 2026 J. Electrochem. Soc. 173 090505
Electrolyte motion induced salt inhomogeneity (EMSI) has been suggested as a novel ageing mechanism occurring in lithium-ion batterys (LIBs). Electrolyte motion refers to the movement and flow of electrolyte within LIBs as a result of active material (AM) expansion and contraction during operation. EMSI is suspected to cause capacity loss and the occurrence of in-plane salt concentration gradients in the electrolyte. To depict the mechanism and deepen understanding of its impact on lithium-ion battery (LIB) performance, we present a nodal-analysis-based pseudo-3D simulation framework that allows efficient computation of the interplay between battery operation and electrolyte flow, utilising equivalent circuit model elements. Using the presented framework, we demonstrate that in-plane concentration gradients and extrema form, even in small-format cells. The resulting concentration extrema lead to conductive and kinetic impediments that reduces the state-of-lithiation window in certain electrode areas, causing the capacity loss. Beyond that, we see in the mid-term an accumulation of Li+ in the electrolyte and in the long-term an accumulation of Li in the AM toward the axial middle of the cell. In sum, the presented framework demonstrates a convenient approach to depict fluid dynamics of the electrolyte in LIBs, motivating further research on electrolyte motion induced salt inhomogeneity (EMSI) and its consequences.
figure placeholder
Masanobu Chiku et al 2026 J. Electrochem. Soc. 173 090503
Two-dimensional metal carbides called MXenes have ability for reversible charge and discharge with the multivalent metal ions. However, the discharge capacity of Ti3C2Tx, the most common type of MXene, was very limited ∼10 mAh g−1 as the positive electrode active material for rechargeable aluminum batteries (RABs). In this study, we investigated the in situ electrochemical activation process of Ti3C2Tx MXene in sulfone-based electrolytes with varying aluminum coordination states. Comprehensive electrochemical, spectroscopic, and structural analyses revealed that only the AlCl3–dimethyl sulfone (DMSO2) electrolyte enabled the progressive enhancement of redox activity through repeated potential cycling. NMR and Raman spectroscopy indicated the dominant formation of solvated Al(DMSO2)33+ species, while X-ray diffraction showed a distinct expansion of the MXene interlayer spacing from 1.23 nm to 1.52 nm after activation. These results demonstrate that the in situ activation originates from the reversible intercalation of strongly solvated Al3+ complexes, which dynamically widen the interlayer gap without significant structural degradation. Although the discharge capacity (∼100 mAh g−1) remains moderate, this study elucidates the key role of electrolyte-derived solvation structures in enabling redox activation of MXene materials, offering mechanistic insights for the rational design of aluminum battery electrodes.
figure placeholder
Henrique D. Minami et al 2026 J. Electrochem. Soc. 173 093502
Aryl diazonium electrografting enables robust covalent bonding of organic molecules to metallic and carbon-based substrates. Despite its widespread use, the influence of specific functional groups on the electrode–electrolyte interfacial response remains insufficiently understood. Once grafted, these molecules alter the electronic environment of the substrate, affecting interfacial stability, charge transfer, and interactions with the surrounding medium. On conductive metals, organic layers have often been reported to increase electron-transfer resistance, and forming stable monolayers remains challenging due to the high reactivity of N≡N+ precursors. Here, we combine quantum-chemistry calculations with impedance spectroscopy to investigate the interfacial behavior of 4-aminophenyl and 4-nitrobenzene groups grafted onto Au surfaces. Experimentally, 4-aminophenyl grafting decreases charge-transfer resistance, while 4-nitrobenzene increases it. Theoretical analysis supports these observations by revealing differences in charge distribution at the interface, adsorption geometries, and bonding strength, clarifying the relative stability and electronic influence of each linker. Conductance is also evaluated in multiple electrode configurations, including systems where carbon nanotubes are linked to gold substrates. This combined theoretical–experimental approach demonstrates how atomistic analysis could guide the design of improved conductive interfaces and highlights the importance of quantum-chemical methods for tailoring molecular anchoring and optimizing nanoscale electronic device performance.
figure placeholder
Primya T. et al 2026 J. Electrochem. Soc. 173 097502
Cancer remains one of the leading causes of mortality worldwide, where early-stage detection significantly improves therapeutic outcomes and survival rates. However, conventional diagnostic modalities such as imaging systems and histopathological examination are often costly, infrastructure-intensive, and inaccessible in resource-constrained healthcare environments, limiting timely screening. To address this gap, this study proposes the Affordable Electrochemical Sensor Platform (AESP)—a cloud-integrated conceptual framework designed to enable rapid and point-of-care cancer biomarker screening in low-resource settings. The AESP architecture integrates a miniaturized electrochemical sensing module with a portable acquisition interface and cloud-based analytical pipeline for biomarker signal interpretation. Rather than representing a physically fabricated device, the platform is currently presented as an architectural and computational model that outlines theoretical signal acquisition, preprocessing, and cloud-assisted decision workflows. The system emphasizes affordability, scalability, and modularity for distributed healthcare deployment. Performance was assessed using public clinical datasets to simulate biomarker classification and signal discrimination, reflecting theoretical predictive capability rather than physical electrochemical testing. Claims on reproducibility, stability, and biological resistance are based on computational modeling, not wet-lab validation. The AESP framework shows feasibility for connecting centralized diagnostics with decentralized screening via cloud analytics. Experimental validation is pending, but it offers a scalable, cost-effective model for cancer screening in underserved regions.
figure placeholder
Maria Shafique et al 2026 J. Electrochem. Soc. 173 090501
This research article focuses on the facile fabrication of pristine cerium oxide (p-CeO2) and platinum-doped cerium oxide (Pt-CeO2) electrocatalyst via chemical reduction method to achieve enhanced specific capacitance for supercapacitor applications in energy storage devices. X-ray diffraction and Scanning Electron Microscopy describe the crystallite size 22 nm of porous heterostructure Pt-CeO2. Electrochemical investigations of the synthesized Pt-CeO2 showed excellent performance as an electrode material for supercapacitor devices. The Pt-CeO2 electrode showed a specific capacity of 542.5 C g−1 and a specific capacitance of 1550 F g−1 at a current density of 0.5 A g−1 compared to p-CeO2 (51 C g−1 and 145 F g−1, respectively). The material also depicts the minimum charge transfer resistance of 618.42 mΩ. The kinetic analysis (b = 0.62, R2 = 0.98) was performed to clarify the super capacitive charge storage mechanism, indicating that the process was predominantly diffusion control. The Pt-CeO2 exhibited remarkable diffusive contributions of 82% and capacitive contributions of 18% at a scan rate of 5 mV s−1. The electrode showed significant cycling stability, retaining 97.16% of its original capacitance after 5000 Galvanic charge discharge cycles (GCD).
figure placeholder
Xinrui Wang et al 2026 J. Electrochem. Soc. 173 082501
The development of environmentally benign, cyanide-free silver electroplating technologies to produce high-quality deposits and replace highly toxic cyanide-based processes is a major research focus in the field. Additives play a vital role in controlling electrodeposition behavior and coating properties. To optimize both bath performance and coating characteristics, research on additives has progressed from studying single-component mechanisms to the systematic design of composite additive systems based on theoretical and experimental foundations. These developments have enabled cyanide-free silver plating not only to match but also to exceed traditional cyanide-based processes in key performance metrics, such as brightness, leveling ability, and adhesion. However, in industrial applications, cyanide-free systems continue to encounter substantial engineering challenges, including the long-term stability of plating solutions, the need for viable operational windows across a wide range of current densities, and the comprehensive costs associated with wastewater treatment. This review systematically summarizes recent advances in cyanide-free silver plating, with a focus on the mechanisms, classification, and research trends of additives. Current challenges in practical applications are also discussed, aiming to provide a theoretical basis and strategic guidance for the development of high-performance, environmentally friendly multifunctional additives.
figure placeholder
Highlights
Focus on the Mechanism of Action and Classification of Additives in Cyanide-Free Silver Plating Systems.
Comprehensively examine single-component and composite additive systems, with particular emphasis on elucidating complex interfacial phenomena within formulations.
Identified the key challenges encountered during the use of cyanide-free silver plating additives.
Gerard J. B. Agravante and Jeff T. Gostick 2026 J. Electrochem. Soc. 173 074507
The parallel electrochemical reactions of platinum dissolution and oxide formation are key reactions that govern surface area loss and subsequent catalyst degradation in polymer electrolyte membrane (PEM) fuel cells. Studying these mechanisms through physics-based approaches is critical for understanding catalyst degradation and for developing more durable fuel cells. This review summarizes advances in physics-based modeling of platinum dissolution and oxidation, presenting three of the most widely used frameworks: the Darling-Meyers, Holby-Morgan, and Rinaldo-Stumper-Eikerling models. These models form the conceptual foundation for many subsequent studies on platinum dissolution and oxidation, and this review examines how recent work has expanded upon these frameworks to illustrate the conceptual evolution of physics-based degradation models. Additional models proposed in the literature are also discussed as alternative approaches that represent newer and emerging directions in modeling platinum degradation. Finally, the review compares the capabilities and limitations of existing models and highlights emerging trends and potential directions for future model development. This review aims to provide a focused guide for researchers developing next-generation physics-based catalyst degradation models for PEM fuel cells.
figure placeholder
Francisco J. Guerrero-Gonzalez et al 2026 J. Electrochem. Soc. 173 072502
Molten Salt Electrolysis (MSE) is a process for extracting oxygen and metals from planetary regolith, contributing to decreasing launch masses and enabling sustainable long-term exploration of the Moon and Mars. MSE offers unique advantages: it tolerates heterogeneous oxide feeds while operating at significantly lower temperatures than alternative thermo- and electro-chemical processes, using a minimal amount of consumables. Two principal MSE technologies, Hall–Héroult electrolysis and the FFC process, are evaluated in terms of their relevance to the space resources field. Drawing on terrestrial heritage, technology gaps that emerge when these processes are translated into extraterrestrial environments are identified. Both technologies are examined in detail across parameters central to regolith processing: anode material, operating temperature, electrolyte composition, feedstock pretreatment, oxide solubility, cathode material, reduction throughput, faradaic efficiency, and the composition of both metallic and gaseous products. This integrated assessment ultimately allows the identification of critical development priorities ranging from anode and cathode durability to electrolyte lifetime and extraterrestrial reactor adaptations, guiding and accelerating the advancement of MSE towards practical off-Earth implementation.
figure placeholder
Highlights
Molten Salt Electrolysis extracts metals and oxygen from planetary regolith.
Connects mature terrestrial electrochemical routes to regolith processing.
Reviews two decades of research on electrometallurgy for space applications.
Evaluates Hall-Héroult and FFC processes across key performance criteria.
Research gaps include space environment adaptation and electrode and bath lifetime.
Ashish Kakoria et al 2026 J. Electrochem. Soc. 173 073502
This review discusses the events of acid mist production at the final stage of hydrometallurgical metal production in copper electrowinning. This review entails the complete process of oxygen bubble generation on the anode and its stages to form into acid mist at the surface of the electrolyte. When these bubbles burst at the solution/air interface, fine acid-containing liquid droplets become airborne and spread throughout the workspace, with the impact factors suppressing the acid mist produced during the process. Also, parameters such as bubble size, temperature, surface tension, and visco-elasticity also affect the acid mist. Sulfuric acid affects the tank houses causes structure deterioration and considerable health effects on employees because sulfuric acid is a carcinogen and costs the industry millions of dollars annually. A theoretical approach with a CFD model is discussed to reduce the acid mist. Furthermore, several other means of suppressing acid mist are shown and discussed, such as the use of surfactants, balls, and hoods. The occupational exposure limits in various nations and the generally used methods of sampling acid mist are described. This review provides a comprehensive understanding of the whole electrowinning process and ways to suppress the acid mist produced during the process.
figure placeholder
Highlights
Electrochemical origin of sulfuric acid mist in copper electrowinning is systematically reviewed
Oxygen bubble nucleation, growth, and bursting mechanisms governing mist formation are analyzed
Effects of physicochemical parameters on acid mist generation and dispersion are critically discussed
CFD-based theoretical approaches for acid mist reduction are summarized
Industrial mitigation strategies and global occupational exposure standards are evaluated
Barbara Thiele et al 2026 J. Electrochem. Soc. 173 064509
Effective water management in polymer electrolyte fuel cells (PEFCs) critically depends on the wettability of gas diffusion layers (GDLs), which controls capillary-driven liquid transport. While hydrophobic coatings (like polytetrafluoroethylene, PTFE) are widely used to tailor wettability, detecting and quantifying their distribution within the complex porous structure remains challenging. Wettability characterization serves as an indirect proxy but is complicated by the coupled effects of pore morphology and surface chemistry, even though effective contact angles remain a required input for capillary transport models. This work summarizes experimental and modeling approaches for GDL wettability assessment. Conventional techniques (sessile drop, Wilhelmy plate) provide surface-level information, whereas bulk methods (Washburn, capillary pressure-saturation (pc–S) curves, capacitance) capture macroscopic behavior but lack spatial resolution. Advanced imaging-based methods, such as environmental scanning electron microscopy and X-ray tomography with image-based contact angle analysis, enable 3D insight yet remain resource-intensive. Furthermore, the widespread concept of “mixed wettability,” attributed to inhomogeneous PTFE coating, is examined and the influence of pore geometry is highlighted, supported by pore-network modeling approaches. Together, these approaches reveal how pore geometry and surface chemistry jointly define GDL wettability.
figure placeholder
William Lvovich et al 2026 J. Electrochem. Soc. 173 082502
This study investigates aqueous electrolytes for efficient iron electrodeposition directly from Fe3+ at ambient temperature. Concentrated lithium chloride (LiCl) based electrolytes were found to stabilize the Fe3+ in the electrolyte, suppress parasitic hydrogen evolution and suppress redox shuttling of intermediate Fe2+, thus promoting efficient iron electrodeposition. These attributes collectively enabled high Coulombic efficiencies (>75%) at high current densities (>200 mA cm−2). Furthermore, detailed investigation revealed that increasing the LiCl concentration increased the electrolyte viscosity and decreased the Fe2+ out-diffusion rate while also suppressing the activity of water. Sufficiently high LiCl concentrations (>7 M) thus provide a window of current densities in which elevated Coulombic efficiencies can be achieved. Overall, this paper develops an understanding of the effects of electrolyte composition on the competition between Fe electrodeposition, hydrogen co-evolution, and intermediate Fe2+ out-diffusion for efficient Fe metal electrosynthesis.
figure placeholder
Riccardo Candeago et al 2025 J. Electrochem. Soc. 172 113503
Production of volatile fatty acids (VFAs) from biomass fermentation is a promising pathway for sustainable chemical production. A central challenge is cost-effective recovery and fractionation of VFAs from fermentation broths. Redox-mediated electrodialysis (redox-ED) can be an energy efficient technique for VFA separation, replacing water splitting with redox-reactions. However, the low selectivity between VFAs of different lengths (separation factors <3) obtained using commercially-available anion-exchange membranes limits the final product purity for industrial use. Cascaded operation can overcome these material limitations and increase product purity, yet multistage redox-ED can be complex and has not been extensively explored for selective ion separations. Here, we developed a physics-based model of redox-ED using finite elements to simulate the separation of a ternary carboxylic acid feed (propionic, butyric, and hexanoic) and evaluated five different multistage architectures including series, parallel, co-current, and counter-current setups. The single-stage, two-dimensional steady-state model captured the effect of the flow-field, concentration, and potential gradients, yielding reasonable agreement with experimental data. We then investigated the tradeoff between number of stages, product purity, and productivity, and assessed the impact of membrane selectivity. In sum, cascaded redox-ED with reflux simultaneously improved product purity and productivity, while series or equivalent single-stage architectures presented a tradeoff between VFA purity and productivity.
figure placeholder
Maria Kelly et al 2025 J. Electrochem. Soc. 172 046503
Voltammetric measurements of electrochemical CO2 reduction reaction (CO2RR) selectivity on rotating ring disk electrodes (RRDE) are a rapid and sensitive method for quantifying an electrocatalyst’s selectivity, i.e. faradaic efficiency (FE). This method has been applied to polycrystalline Au electrocatalysts where a Au disk electrode catalyzes both the CO2RR and hydrogen evolution reaction while the concentric Au ring electrode selectively senses CO by oxidizing CO back to CO2. Such measurements enabled fundamental mechanistic studies but suffer from poor inter-laboratory reproducibility. This work identifies causes of variability in RRDE selectivity measurements by comparing protocols with different electrochemical methods, reagent purities, and glassware cleaning procedures. We observed FECO decrease by 56% during 5 min chronoamperometry measurements, a phenomenon that is not readily apparent in voltammetric scans due to their dynamic nature. Electroplating of electrolyte impurities onto the disk and ring surfaces were identified as a major contributor to Au deactivation. Additionally, the oxygen reduction reaction may lead to higher disk currents in inadequately purged electrolytes, causing an apparent underestimation of FECO at low overpotentials. Lastly, we propose operational bounds for CO2RR selectivity measurements on Au using the RRDE method and provide suggestions on steps for improving the accuracy of this technique.
figure placeholder
Anar Badalbayli et al 2025 J. Electrochem. Soc. 172 032508
This study explores chloride molten salt electrolysis (CMSE) as a promising route for energy-efficient iron metal (Fe) production. Moderate temperature (500 °C) LiCl-KCl molten salts offer excellent thermodynamic stability, high ionic conductivity and diffusivity, and high solubility for FeCl3, thereby enabling efficient Fe metal extraction at high electrowinning rates. Here, we demonstrate the two essential steps for converting taconite ore into Fe metal. First, Fe2O3 from taconite pellets was selectively leached in HCl yielding a high-purity FeCl3 aqueous solution, while the gangue components settled at the bottom. Then, anhydrous FeCl3 was electrolyzed in a LiCl-KCl eutectic molten salt at 500 °C at high current density (1 A cm−2) and at high Coulombic efficiency (>85%). Analysis of the electrowon Fe deposits revealed dendritic structures with purity of >99 wt%, which could be further improved to nearly 100 wt% through arc re-melting. CMSE offers low specific energy consumption (3.7 kWhr kg−1), competitive with H2-DRI and other electrolytic approaches being pursued globally. Our findings underscore the potential of CMSE as an energy-efficient route for electrosynthesis of Fe metal.
Guoxin Li et al 2025 J. Electrochem. Soc. 172 026501
Cathode-electrolyte interphase (CEI) is critical for inhibiting the cathode degradation to maintain cell life. However, the evolution of the CEI is still unclear due to its complex and slow dynamic process. Here we used scanning electrochemical microscopy (SECM) for in situ investigation of CEI formation process on LiFePO4 cathode. Feedback images and probe scan curves showed a heterogeneous passivation that was gently generated on the LiFePO4 particles during both charging and discharging. Besides, a LiFePO4 composited electrode was also used to investigate the CEI formation to simulate the condition of real battery system. The composited cathode does not show obvious CEI formation within first two cycles. The SECM results between the pristine LiFePO4 particles and the composited LiFePO4 indicated the dynamic accumulation of CEI, which is influenced by the ability to charge transfer kinetics of cathode materials. This approach provided a feasible consideration for the connections between the dynamic evolution of the CEI and changes in charge transfer capability of cathode during cycling.
figure placeholder
Highlights
In-situ investigation of cathode-electrolyte interphase formation.
The evolution of native active material and composite slurry were compared.
The electrochemical activity change upon cathode cycling are analysed in situ.
The influence of the charge transfer capability upon CEI generation is revealed.
Kongkanand et al
Mathematical modeling of performance and durability plays a central role in developing electrochemical devices, where complex trade-offs between functionality, longevity, and cost are frequently done. To shorten learning cycles when developing a proton exchange membrane fuel cell (PEMFC) to last tens of thousands of hours, accelerated stress tests (ASTs) are commonly used. However, this can mischaracterize degradation mechanisms leading to unpreferred outcomes. In this work, we investigate platinum (Pt) surface area loss and carbon support degradation under non-accelerated, realistic operating conditions. Leveraging a unique dataset spanning thousands of hours of durability testing, we developed semi-empirical models to project the operational lifespans of both Pt and carbon components. The models were then applied to evaluate the viability of PEMFCs across a diverse range of potential fuel cell applications.
Han et al
To recover Pr aided by Cu electrode, LiF-LiCl melt was chosen as electrolyte to study the electrochemical behavior of Pr(Ⅲ) on W and Cu film electrodes using a sequence of electrochemical techniques. The results of CV, SWV, and CP revealed that on W electrode, the electrode reaction of Pr(Ⅲ) proceeded in a single-step three-electron process that was diffusion-controlled and reversible. The dynamic properties were estimated by CV and LP. The peak potentials of Cu-Pr intermetallic compounds were found to be more positive than that of Pr(Ⅲ) due to the underpotential deposition. Moreover, and apr of Pr in two-phase equilibrium as well as , and of Cu-Pr alloy compounds were calculated by OCP. Furthermore, the electrolytic extraction of Pr was explored assisted by Cu electrode, the products were characterized by XRD, SEM-EDS. The results showed that Cu6Pr, Cu5Pr and Cu2Pr were prepared at different applied potentials. The recovery rate and efficiency of Pr at different durations were estimated by measuring the content of Pr(Ⅲ) using ICP-AES. With prolonging the electrochemical recovery time, the recovery rate declined while the recovery efficiency raised. After 16 h of electrolysis at -1.98 V, the recovery efficiency could reach 95.0%.
Thackeray et al
This narrative summarizes, in retrospect, the enormous and quietly-conducted contribution of Dr. Johan Coetzer, a South African scientist and entrepreneur, to the discovery, development and implementation of the high temperature sodium – metal chloride ‘ZEBRA’ battery (Na/-Al2O3, NaAlCl4/MCl2 (M=Ni, Fe)). This research activity was initiated in the mid 1970’s at the Council for Scientific and Industrial Research (CSIR) in South Africa and subsequently developed primarily in partnership with the Atomic Energy Research Establishment (AERE, Harwell, UK), Beta R&D (Derby, UK) and Daimler Benz (Germany), before being transferred to industry worldwide for electric vehicle and energy storage applications, albeit at a relatively low production rate. Coetzer’s wide-ranging, innovative and, at times, unconventional scientific approach also laid the foundation for the discovery and implementation of manganese-based spinel and layered metal oxide cathode materials for the Li-ion battery industry.
Diaz et al
Silicon anodes in lithium-ion batteries have experienced significant advances that mitigate degradation from cell cycling; however, challenges related to poor calendar life persist. Cycle life improvements have largely been due to the development of nano-silicon materials which greatly increases electrode surface area. This study systematically investigates the influence of electrode-level porosity on calendar life performance of silicon anodes. The anodes were extensively characterized and subjected to accelerated calendar aging protocols in coin cells and more typical calendar aging testing in pouch cells. Results from the coin cell aging tests demonstrated a correlation between increasing anode porosity and increasing capacity loss during accelerated calendar aging. This correlation was much less pronounced in subsequent testing of larger format pouch cells with longer duration, more representative calendar aging protocols. These findings provide critical insights into the ability to enhance calendar lifetimes through the modification of silicon anode architecture design.
Yang et al
Through-mask electrochemical micromachining (TMEMM) is a key technique for fabricating metal microstructures. However, manipulating the electric field to achieve various machining profiles remains a persistent challenge. To address this limitation, this paper introduces a sacrificial electrode into induction electrode TMEMM (IETMEMM) for the first time and proposes a novel method called sacrificial electrode-assisted IETMEMM (SE-IETMEMM). The sacrificial electrode is placed on the photoresist and is electrically connected to the workpiece through the regulating resistor. By adjusting the resistance, the potential of the sacrificial electrode is altered, enabling precise manipulation of the electric field distribution on the machined surface. Specifically, as the resistance increases, the electric field distribution evolves from center concentration to a uniform distribution and subsequently to edge concentration. This evolution is experimentally verified using the machining profiles of gear patterns. At a resistance of 0 Ω, the fabricated specimen exhibits a shallow edge and a deep center, indicating high localization with an etching factor (EF) of 10.3. At a resistance of 0.1 Ω, the profile becomes uniformly flat, with a non-uniformity (Nu) of only 12.2%. At a resistance of 1 Ω, the fabricated specimen exhibits an island-like profile.
Trending on Altmetric
Marcel Rogge et al 2026 J. Electrochem. Soc. 173 090505
Electrolyte motion induced salt inhomogeneity (EMSI) has been suggested as a novel ageing mechanism occurring in lithium-ion batterys (LIBs). Electrolyte motion refers to the movement and flow of electrolyte within LIBs as a result of active material (AM) expansion and contraction during operation. EMSI is suspected to cause capacity loss and the occurrence of in-plane salt concentration gradients in the electrolyte. To depict the mechanism and deepen understanding of its impact on lithium-ion battery (LIB) performance, we present a nodal-analysis-based pseudo-3D simulation framework that allows efficient computation of the interplay between battery operation and electrolyte flow, utilising equivalent circuit model elements. Using the presented framework, we demonstrate that in-plane concentration gradients and extrema form, even in small-format cells. The resulting concentration extrema lead to conductive and kinetic impediments that reduces the state-of-lithiation window in certain electrode areas, causing the capacity loss. Beyond that, we see in the mid-term an accumulation of Li+ in the electrolyte and in the long-term an accumulation of Li in the AM toward the axial middle of the cell. In sum, the presented framework demonstrates a convenient approach to depict fluid dynamics of the electrolyte in LIBs, motivating further research on electrolyte motion induced salt inhomogeneity (EMSI) and its consequences.
figure placeholder
Kentaro U. Hansen et al 2026 J. Electrochem. Soc. 173 084505
An XY segmented cell was developed for low temperature PEM water electrolysis (PEMWE). The system can assess the local performance by enabling in situ measurements of spatial currents and impedances. In this work, we show through experiments, as well as through modelling work, that the porous transport layer (PTL) must be segmented to eliminate crosstalk. Accurate measurements are only possible when crosstalk is fully eliminated. The XY segmented cell is applied to a case study characterizing the impact of a PTL platinum coating void on spatial performance. The localized performance impact of the coating void is found to be orientation specific: coating voids facing the catalyst layer reduce performance significantly more than coating voids facing the flow field. The results suggest that the tolerances for PTL coating uniformity can be lower at the side facing the flow field. The work showcases the feasibility of the XY segmented cell for impact assessment studies. The presented XY segmented cell enables the characterization of spatial phenomena in PEMWE devices and is envisioned to support modeling efforts and the investigation of manufacturing related tolerances for mass produced PEMWE devices.
figure placeholder
Highlights
Electronic crosstalk between flow field segments can cause major measurement errors
Shunt resistor choice directly impacts degree of crosstalk between segments
Crosstalk between segments eliminated by segmenting the porous transport layer
Standard electrolyzer configuration assessed using segmented cell
Voids in porous transport layer coating affect performance depending on orientation
William Lvovich et al 2026 J. Electrochem. Soc. 173 082502
This study investigates aqueous electrolytes for efficient iron electrodeposition directly from Fe3+ at ambient temperature. Concentrated lithium chloride (LiCl) based electrolytes were found to stabilize the Fe3+ in the electrolyte, suppress parasitic hydrogen evolution and suppress redox shuttling of intermediate Fe2+, thus promoting efficient iron electrodeposition. These attributes collectively enabled high Coulombic efficiencies (>75%) at high current densities (>200 mA cm−2). Furthermore, detailed investigation revealed that increasing the LiCl concentration increased the electrolyte viscosity and decreased the Fe2+ out-diffusion rate while also suppressing the activity of water. Sufficiently high LiCl concentrations (>7 M) thus provide a window of current densities in which elevated Coulombic efficiencies can be achieved. Overall, this paper develops an understanding of the effects of electrolyte composition on the competition between Fe electrodeposition, hydrogen co-evolution, and intermediate Fe2+ out-diffusion for efficient Fe metal electrosynthesis.
figure placeholder
Megan Diaz et al 2026 J. Electrochem. Soc.
Silicon anodes in lithium-ion batteries have experienced significant advances that mitigate degradation from cell cycling; however, challenges related to poor calendar life persist. Cycle life improvements have largely been due to the development of nano-silicon materials which greatly increases electrode surface area. This study systematically investigates the influence of electrode-level porosity on calendar life performance of silicon anodes. The anodes were extensively characterized and subjected to accelerated calendar aging protocols in coin cells and more typical calendar aging testing in pouch cells. Results from the coin cell aging tests demonstrated a correlation between increasing anode porosity and increasing capacity loss during accelerated calendar aging. This correlation was much less pronounced in subsequent testing of larger format pouch cells with longer duration, more representative calendar aging protocols. These findings provide critical insights into the ability to enhance calendar lifetimes through the modification of silicon anode architecture design.
Jack Guo et al 2026 J. Electrochem. Soc. 173 084504
Tandem catalysis involves two or more catalysts arranged in proximity within a single reaction vessel, with the aim of synergistically aligning the catalysts’ reaction pathways to maximize overall system performance. This study presents a proof of concept showing the integration of continuum transport modeling with design optimization in a simplified two-dimensional flow reactor setup for electrochemical CO2 reduction. Ag catalysts provide the CO2 ⟶ CO reaction capability, and Cu catalysts provide the CO ⟶ high-value products reaction capability. Given a set of input parameters, the optimization algorithm uses adjoint methods to modify the Ag/Cu surface patterning in order to maximize the current density toward high-value products, such as ethylene. The optimized designs yield significant performance enhancement especially at more negative applied voltages (i.e., stronger surface reactions) and for larger numbers of patterning sections. For an applied voltage of −1.7 V vs. SHE, the 12-section optimized design increases the current density toward ethylene by up to 65% compared to the unoptimized 2-section design. For the optimized cases, observed differences in the production and consumption of CO (the key intermediate species) and minimized zones of low CO reactant surface concentration on Cu sections explain the improved reactor performance.
figure placeholder
Nancy N. Kariuki et al 2026 J. Electrochem. Soc. 173 084503
One of the main degradation mechanisms of hydrous iridium oxide acidic oxygen evolution reaction (OER) catalysts is dissolution and loss into the acidic membrane. While degradation models have been proposed, there is a gap in understanding the potential and time dependence of the iridium dissolution reaction and its mechanistic underpinnings. In this work, Ir dissolution rates measured as a function of time and potential via time-resolved inductively-coupled plasma mass spectrometry (ICP-MS) in aqueous acidic electrolyte are used to establish a mathematical model for Ir dissolution. The mathematical model is generated using proposed formation and dissolution reactions for Ir species. Through comparison with the ICP-MS data and existing information on the potential-dependent Ir phase, we find that the potential and time-dependence of dissolution can be modeled as dissolution of an oxide phase, here represented as IrO2, with potential dependent kinetics and formation of a passivating species, a process with a rate-limiting step that is not potential dependent. This understanding of the potential dependence of dissolution and passivation kinetics using aqueous electrolyte half-cell measurements can be used to predict the degradation of Ir oxide in operating energy conversion devices relying on the OER, such as proton-exchange membrane water electrolyzers.
figure placeholder
Harsh Srivastav et al 2026 J. Electrochem. Soc. 173 084502
Prediction of the catalyst-activated carbon particle sizes of fuel-cell inks remains a critical challenge in enhancing the performance and durability of fuel cells. The performance and structural integrity of the catalyst layers in the cell depend not only on the properties of the ionomer, but also on the carbon supports that host the catalyst. To investigate how these carbon aggregate structures form, we present a model that calculates the cooperative size distributions of ionomer and carbon aggregates in various water/alcohol mixtures and compares those results to available experimental data. Aggregation of both the suspended ionomers and the carbon particles is interwoven as the carbon aggregation depends heavily on the ionic strength of its environment, namely protons dissociating from the ionomer’s sulfonic-acid-group side chains. We demonstrate that the surrounding mixed solvent as well as NafionTM binder concentration strongly influence the degree of aggregation for carbon particles. The validated model provides a framework for exploring aggregation under a wide range of conditions for various electrochemical-technology inks.
figure placeholder
Marco-Tulio F. Rodrigues et al 2026 J. Electrochem. Soc.
Silicon-containing lithium-ion batteries can exhibit capacity gain early in life, which makes forecasting future cell behavior difficult. We have observed these anomalous trends even in conditions where known mechanisms, such as overhang equalization and excessive electrolyte oxidation, are unlikely to be significant. Here, we combine simulations and experiments to analyze four cases that can produce increased capacity in Si cells. Three of these pathways relate to “break-in” processes, where improved mass and charge transport can lead to increased access to active electrode domains and decreased cell impedance. The fourth case occurs at high levels of prelithiation, when the positive electrode (PE) is completely replenished with Li+ at the end of cell discharge. We show that the commonality among these mechanisms is that the underlying transformations change the potentials experienced by electrodes at the end of half-cycles, increasing the Li+ inventory available to the cell. A quantitative framework to describe these effects is presented, enabling these ideas to be extended to other battery systems.
James Nicholas Ashley et al 2026 J. Electrochem. Soc.
Dilute-solution models based on the Nernst–Planck equations are commonly used for describing ion transport in flow-battery electrolytes, yet they fail to accurately capture the behaviour at the high ion concentrations typical of these systems. To overcome this limitation, we develop a concentrated-solution theory model for a multispecies electrolyte using the Stefan–Maxwell framework. Our focus is the ternary aqueous electrolyte employed in soluble-lead flow batteries, consisting of a solution of lead methanesulfonate, Pb(CH3SO3)2, in methanesulfonic acid, CH3SO3H. Molecular dynamics simulations and laboratory experiments are combined to fully parametrise this model. A Green–Kubo approach is used to compute Stefan–Maxwell diffusivities from MD data (see [D. R. Wheeler and J. Newman, J. Phys. Chem. B, 108, 18353 (2004)]). In combination with experiment, this leads to analytic expressions for density, kinematic viscosity, and the six independent Stefan-Maxwell diffusivities describing the system. Agreement between the molecular dynamics simulations and experiments is achieved using only three physically justified fitting parameters. The methodology provides a rigorous framework for parametrising continuum-physics models of multispecies electrolytes derived from concentrated solution theory and is readily adaptable to other electrochemical sys-
tems.
Baichen Liu et al 2026 J. Electrochem. Soc. 173 080503
Redox flow batteries (RFBs) are promising for large-scale energy storage, but their widespread deployment is limited by elevated capital costs. Advancing porous electrode to achieve high power density is an effective strategy for reducing the system-level costs. Conventional fibrous electrodes are not originally designed for RFBs, resulting in low surface area and suboptimal microstructure. Recently, non-solvent induced phase separation (NIPS) has emerged as a versatile method for fabricating non-fibrous electrodes with engineered architectures. Building on our previous NIPS work, we introduce a nanoparticle-assisted templating strategy to achieve multimodal pore networks. CaCO3 nanoparticles are incorporated into the polymer solution and subsequently removed to template mesopores within the scaffold. By varying the CaCO3 loading from 2.5 wt% to 15 wt%, we systematically evaluate the effects on solution rheology, microstructure, pressure drop, and electrochemical performance. The resulting electrodes exhibit trimodal pore networks, enabling high permeability, enhanced electrochemical surface area, and efficient electrolyte transport. Electrochemical testing in vanadium RFBs shows a significant increase in peak power density, with optimal performance achieved at 5 wt% loading (757 mW cm−2). These results highlight the benefits of multimodal pore microstructures in RFB electrodes and open a pathway for electrode engineering applicable to other electrochemical technologies.
figure placeholder
Wolfgang Schmickler and Sergio Trasatti 2006 J. Electrochem. Soc. 153 L31
We compare the adsorption energies calculated by Nørskov et al. with data derived from experimental values; except for Ni and Co, which absorb hydrogen strongly, there is a linear relation. We discuss the model proposed by these authors in the light of extensive previous work and of experimental data and find it overly simplistic.
J. K. Nørskov et al 2005 J. Electrochem. Soc. 152 J23
A density functional theory database of hydrogen chemisorption energies on close packed surfaces of a number of transition and noble metals is presented. The bond energies are used to understand the trends in the exchange current for hydrogen evolution. A volcano curve is obtained when measured exchange currents are plotted as a function of the calculated hydrogen adsorption energies and a simple kinetic model is developed to understand the origin of the volcano. The volcano curve is also consistent with Pt being the most efficient electrocatalyst for hydrogen evolution. © 2005 The Electrochemical Society. All rights reserved.
E. Peled and S. Menkin 2017 J. Electrochem. Soc. 164 A1703
The Solid-Electrolyte-Interphase (SEI) model for non-aqueous alkali-metal batteries constitutes a paradigm change in the understanding of lithium batteries and has thus enabled the development of safer, durable, higher-power and lower-cost lithium batteries for portable and EV applications. Prior to the publication of the SEI model (1979), researchers used the Butler-Volmer equation, in which a direct electron transfer from the electrode to lithium cations in the solution is assumed. The SEI model proved that this is a mistaken concept and that, in practice, the transfer of electrons from the electrode to the solution in a lithium battery, must be prevented, since it will result in fast self-discharge of the active materials and poor battery performance. This model provides [E. Peled, in “Lithium Batteries,” J.P. Gabano (ed), Academic Press, (1983), E. Peled, J. Electrochem. Soc., 126, 2047 (1979).] new equations for: electrode kinetics (io and b), anode corrosion, SEI resistivity and growth rate and irreversible capacity loss of lithium-ion batteries. This model became a cornerstone in the science and technology of lithium batteries. This paper reviews the past, present and the future of SEI batteries.
D. A. Stevens and J. R. Dahn 2000 J. Electrochem. Soc. 147 1271
Electrochemical techniques have been used to study the reversible insertion of sodium into hard‐carbon host structures at room temperature. In this paper we compare these results with those for lithium insertion in the same materials and demonstrate the presence of similar alkali metal insertion mechanisms in both cases. Despite the gravimetric capacities being lower for sodium than lithium insertion, we have achieved a reversible sodium capacity of 300 mAh/g, close to that for lithium insertion in graphitic carbon anode materials. Such materials may therefore be useful as anodes in rechargeable sodium‐ion batteries. © 2000 The Electrochemical Society. All rights reserved.
Roland Jung et al 2017 J. Electrochem. Soc. 164 A1361
Layered LiNixMnyCozO2 (NMC) is a widely used class of cathode materials with LiNi1/3Mn1/3Co1/3O2 (NMC111) being the most common representative. However, Ni-rich NMCs are more and more in the focus of current research due to their higher specific capacity and energy. In this work we will compare LiNi1/3Mn1/3Co1/3O2 (NMC111), LiNi0.6Mn0.2Co0.2O2 (NMC622), and LiNi0.8Mn0.1Co0.1O2 (NMC811) with respect to their cycling stability in NMC-graphite full-cells at different end-of-charge potentials. It will be shown that stable cycling is possible up to 4.4 V for NMC111 and NMC622 and only up to 4.0 V for NMC811. At higher potentials, significant capacity fading was observed, which was traced back to an increase in the polarization of the NMC electrode, contrary to the nearly constant polarization of the graphite electrode. Furthermore, we show that the increase in the polarization occurs when the NMC materials are cycled up to a high-voltage feature in the dq/dV plot, which occurs at ∼4.7 V vs. Li/Li+ for NMC111 and NMC622 and at ∼4.3 V vs. Li/Li+ for NMC811. For the latter material, this feature corresponds to the H2 → H3 phase transition. Contrary to the common understanding that the electrochemical oxidation of carbonate electrolytes causes the CO2 and CO evolution at potentials above 4.7 V vs. Li/Li+, we believe that the observed CO2 and CO are mainly due to the chemical reaction of reactive lattice oxygen with the electrolyte. This hypothesis is based on gas analysis using On-line Electrochemical Mass Spectrometry (OEMS), by which we prove that all three materials release oxygen from the particle surface and that the oxygen evolution coincides with the onset of CO2 and CO evolution. Interestingly, the onsets of oxygen evolution for the different NMCs correlate well with the high-voltage redox feature at ∼4.7 V vs. Li/Li+ for NMC111 and NMC622 as well as at ∼4.3 V vs. Li/Li+ for NMC811. To support this hypothesis, we show that no CO2 or CO is evolved for the LiNi0.43Mn1.57O4 (LNMO) spinel up to 5 V vs. Li/Li+, consistent with the absence of oxygen release. Lastly, we demonstrate by the use of 13C labeled conductive carbon that it is the electrolyte rather than the conductive carbon which is oxidized by the released lattice oxygen. Taking these findings into consideration, a mechanism is proposed for the reaction of released lattice oxygen with ethylene carbonate yielding CO2, CO, and H2O.
Todd M. Bandhauer et al 2011 J. Electrochem. Soc. 158 R1
Lithium-ion batteries are well-suited for fully electric and hybrid electric vehicles due to their high specific energy and energy density relative to other rechargeable cell chemistries. However, these batteries have not been widely deployed commercially in these vehicles yet due to safety, cost, and poor low temperature performance, which are all challenges related to battery thermal management. In this paper, a critical review of the available literature on the major thermal issues for lithium-ion batteries is presented. Specific attention is paid to the effects of temperature and thermal management on capacity/power fade, thermal runaway, and pack electrical imbalance and to the performance of lithium-ion cells at cold temperatures. Furthermore, insights gained from previous experimental and modeling investigations are elucidated. These include the need for more accurate heat generation measurements, improved modeling of the heat generation rate, and clarity in the relative magnitudes of the various thermal effects observed at high charge and discharge rates seen in electric vehicle applications. From an analysis of the literature, the requirements for lithium-ion thermal management systems for optimal performance in these applications are suggested, and it is clear that no existing thermal management strategy or technology meets all these requirements.
Charles Monroe and John Newman 2005 J. Electrochem. Soc. 152 A396
Past theories of electrode stability assume that the surface tension resists the amplification of surface roughness at cathodes and show that instability at lithium/liquid interfaces cannot be prevented by surface forces alone [Electrochim. Acta, 40, 599 (1995)]. This work treats interfacial stability in lithium/polymer systems where the electrolyte is solid. Linear elasticity theory is employed to compute the additional effect of bulk mechanical forces on electrode stability. The lithium and polymer are treated as Hookean elastic materials, characterized by their shear moduli and Poisson’s ratios. Two-dimensional displacement distributions that satisfy force balances across a periodically deforming interface are derived; these allow computation of the stress and surface-tension forces. The incorporation of elastic effects into a kinetic model demonstrates regimes of electrolyte mechanical properties where amplification of surface roughness can be inhibited. For a polymer material with Poisson’s ratio similar to poly(ethylene oxide), interfacial roughening is mechanically suppressed when the separator shear modulus is about twice that of lithium. © 2005 The Electrochemical Society. All rights reserved.
D. A. Stevens and J. R. Dahn 2001 J. Electrochem. Soc. 148 A803
In this paper, we compare the interactions of lithium and sodium with a range of carbon materials in electrochemical cells. Through wide angle in situ X-ray scattering studies, we demonstrate that both lithium and sodium can be inserted into the interlayer space in disordered carbon materials. This insertion process is accompanied by an increase in the interlayer spacing in these materials. Small-angle in situ scattering studies are presented to clearly show the insertion of lithium and sodium into nanopores within disordered hard carbons. We also show that very little, if any, sodium can be inserted into graphitic materials in contrast to the large capacity seen for lithium insertion. © 2001 The Electrochemical Society. All rights reserved.
George E. Blomgren 2017 J. Electrochem. Soc. 164 A5019
This year, the battery industry celebrates the 25th anniversary of the introduction of the lithium ion rechargeable battery by Sony Corporation. The discovery of the system dates back to earlier work by Asahi Kasei in Japan, which used a combination of lower temperature carbons for the negative electrode to prevent solvent degradation and lithium cobalt dioxide modified somewhat from Goodenough's earlier work. The development by Sony was carried out within a few years by bringing together technology in film coating from their magnetic tape division and electrochemical technology from their battery division. The past 25 years has shown rapid growth in the sales and in the benefits of lithium ion in comparison to all the earlier rechargeable battery systems. Recent work on new materials shows that there is a good likelihood that the lithium ion battery will continue to improve in cost, energy, safety and power capability and will be a formidable competitor for some years to come.
Wenchao Sheng et al 2010 J. Electrochem. Soc. 157 B1529
The kinetics of the hydrogen oxidation reaction (HOR) and hydrogen evolution reaction (HER) on polycrystalline platinum [Pt(pc)] and high surface area carbon-supported platinum nanoparticles (Pt/C) were studied in 0.1 M KOH using rotating disk electrode (RDE) measurements. After corrections of noncompensated solution resistance from ac impedance spectroscopy and of hydrogen mass transport in the HOR branch, the kinetic current densities were fitted to the Butler–Volmer equation using a transfer coefficient of 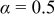 , from which HOR/HER exchange current densities on Pt(pc) and Pt/C were obtained, and the HOR/HER mechanisms in alkaline solution were discussed. Unlike the HOR/HER rates on Pt electrodes in alkaline solution, the HOR/HER rates on a Pt electrode in 0.1 M
, from which HOR/HER exchange current densities on Pt(pc) and Pt/C were obtained, and the HOR/HER mechanisms in alkaline solution were discussed. Unlike the HOR/HER rates on Pt electrodes in alkaline solution, the HOR/HER rates on a Pt electrode in 0.1 M 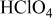 were limited entirely by hydrogen diffusion, which renders the quantification of the HOR/HER kinetics impossible by conventional RDE measurements. The simulation of the hydrogen anode performance based on the specific exchange current densities of the HOR/HER at
were limited entirely by hydrogen diffusion, which renders the quantification of the HOR/HER kinetics impossible by conventional RDE measurements. The simulation of the hydrogen anode performance based on the specific exchange current densities of the HOR/HER at 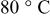 illustrates that in addition to the oxygen reduction reaction cell voltage loss on the cathode, the slow HOR kinetics are projected to cause significant anode potential losses in alkaline fuel cells for low platinum loadings (
illustrates that in addition to the oxygen reduction reaction cell voltage loss on the cathode, the slow HOR kinetics are projected to cause significant anode potential losses in alkaline fuel cells for low platinum loadings (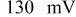 at
at 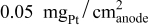 and
and 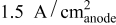 ), contrary to what is reported for proton exchange membrane fuel cells.
), contrary to what is reported for proton exchange membrane fuel cells.
Journal links
ECS Publications
- Journal of the Electrochemical Society
- ECS Journal of Solid State Science and Technology
- ECS Advances
- ECS Sensors Plus
- ECS Transactions
- The Electrochemical Society Interface
- ECS Meeting Abstracts
- ECS Electrochemistry Letters
- ECS Solid State Letters
- Electrochemical and Solid State Letters
- ECS Proceedings Volumes
Journal information
- 1902-present
Journal of The Electrochemical Society
doi: 10.1149/issn.1945-7111
Online ISSN: 1945-7111
Print ISSN: 0013-4651